Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Acetylcholinesterase
Ligand
BDBM10435
Substrate
BDBM8959
Meas. Tech.
Enzyme Inhibition Assay
IC50
900±n/a nM
Citation
More Info.:
Target
Name:
Acetylcholinesterase
Synonyms:
3.1.1.7 | ACES_TETCF | Acetylcholinesterase (AChE) | Acetylcholinesterase precursor | ache
Type:
n/a
Mol. Mass.:
65900.74
Organism:
Tetronarce californica (Pacific electric ray) (Torpedo californica)
Description:
P04058
Residue:
586
Sequence:
MNLLVTSSLGVLLHLVVLCQADDHSELLVNTKSGKVMGTRVPVLSSHISAFLGIPFAEPPVGNMRFRRPEPKKPWSGVWNASTYPNNCQQYVDEQFPGFSGSEMWNPNREMSEDCLYLNIWVPSPRPKSTTVMVWIYGGGFYSGSSTLDVYNGKYLAYTEEVVLVSLSYRVGAFGFLALHGSQEAPGNVGLLDQRMALQWVHDNIQFFGGDPKTVTIFGESAGGASVGMHILSPGSRDLFRRAILQSGSPNCPWASVSVAEGRRRAVELGRNLNCNLNSDEELIHCLREKKPQELIDVEWNVLPFDSIFRFSFVPVIDGEFFPTSLESMLNSGNFKKTQILLGVNKDEGSFFLLYGAPGFSKDSESKISREDFMSGVKLSVPHANDLGLDAVTLQYTDWMDDNNGIKNRDGLDDIVGDHNVICPLMHFVNKYTKFGNGTYLYFFNHRASNLVWPEWMGVIHGYEIEFVFGLPLVKELNYTAEEEALSRRIMHYWATFAKTGNPNEPHSQESKWPLFTTKEQKFIDLNTEPMKVHQRLRVQMCVFWNQFLPKLLNATACDGELSSSGTSSSKGIIFYVLFSILYLIF
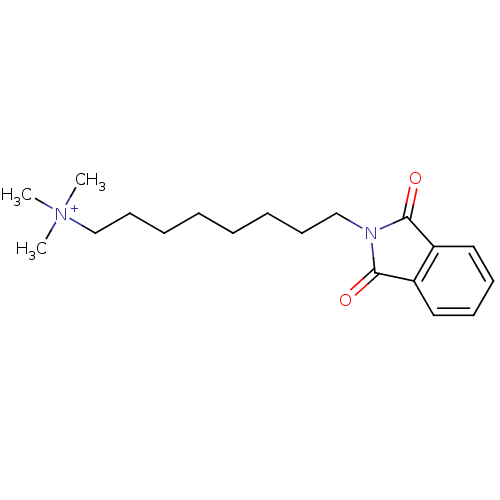
Inhibitor
Name:
BDBM10435
Synonyms:
8-(Phthalimidooctyl)-trimethylammonium bromide | [8-(1,3-dioxo-2,3-dihydro-1H-isoindol-2-yl)octyl]trimethylazanium bromide | galanthamine deriv. 13d
Type:
Small organic molecule
Emp. Form.:
C19H29N2O2
Mol. Mass.:
317.4452
SMILES:
C[N+](C)(C)CCCCCCCCN1C(=O)c2ccccc2C1=O
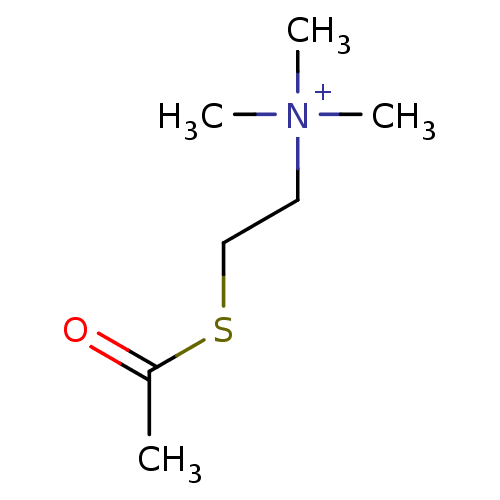
Substrate
Name:
BDBM8959
Synonyms:
(2-Mercaptoethyl)trimethylammonium iodide acetate | ATC | Acetylthiocholine | [2-(acetylsulfanyl)ethyl]trimethylazanium iodide | acetylthiocholine chloride | acetylthiocholine iodide
Type:
Small organic molecule
Emp. Form.:
C7H16NOS
Mol. Mass.:
162.272
SMILES:
CC(=O)SCC[N+](C)(C)C