Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutamate carboxypeptidase 2 [44-750]
Ligand
BDBM17664
Substrate
BDBM17658
Meas. Tech.
Determination of Inhibition Concentration Values (IC50)
pH
7.4±n/a
Temperature
310.15±n/a K
IC50
523000±n/a nM
Citation
 Barinka, C; Rovenská, M; Mlcochová, P; Hlouchová, K; Plechanovová, A; Majer, P; Tsukamoto, T; Slusher, BS; Konvalinka, J; Lubkowski, J Structural insight into the pharmacophore pocket of human glutamate carboxypeptidase II. J Med Chem 50:3267-73 (2007) [PubMed] Article
Barinka, C; Rovenská, M; Mlcochová, P; Hlouchová, K; Plechanovová, A; Majer, P; Tsukamoto, T; Slusher, BS; Konvalinka, J; Lubkowski, J Structural insight into the pharmacophore pocket of human glutamate carboxypeptidase II. J Med Chem 50:3267-73 (2007) [PubMed] Article Target
Name:
Glutamate carboxypeptidase 2 [44-750]
Synonyms:
FOLH | FOLH1 | FOLH1_HUMAN | Folate hydrolase 1 | Folylpoly-gamma-glutamate carboxypeptidase | Glutamate Carboxypeptidase II (GCPII) | Glutamate carboxypeptidase 2 | Membrane glutamate carboxypeptidase | NAALAD1 | PSM | PSMA | Prostate-specific membrane antigen | Prostate-specific membrane antigen (PSMA) | Pteroylpoly-gamma-glutamate carboxypeptidase | mGCP
Type:
Enzyme
Mol. Mass.:
79524.51
Organism:
Homo sapiens (Human)
Description:
The extracellular domain of human glutamate carboxypeptidase II (rhGCPII, amino acids 44-750) was overexpressed in Drosophila Schneider S2 cells and purified to homogeneity.
Residue:
707
Sequence:
KSSNEATNITPKHNMKAFLDELKAENIKKFLYNFTQIPHLAGTEQNFQLAKQIQSQWKEFGLDSVELAHYDVLLSYPNKTHPNYISIINEDGNEIFNTSLFEPPPPGYENVSDIVPPFSAFSPQGMPEGDLVYVNYARTEDFFKLERDMKINCSGKIVIARYGKVFRGNKVKNAQLAGAKGVILYSDPADYFAPGVKSYPDGWNLPGGGVQRGNILNLNGAGDPLTPGYPANEYAYRRGIAEAVGLPSIPVHPIGYYDAQKLLEKMGGSAPPDSSWRGSLKVPYNVGPGFTGNFSTQKVKMHIHSTNEVTRIYNVIGTLRGAVEPDRYVILGGHRDSWVFGGIDPQSGAAVVHEIVRSFGTLKKEGWRPRRTILFASWDAEEFGLLGSTEWAEENSRLLQERGVAYINADSSIEGNYTLRVDCTPLMYSLVHNLTKELKSPDEGFEGKSLYESWTKKSPSPEFSGMPRISKLGSGNDFEVFFQRLGIASGRARYTKNWETNKFSGYPLYHSVYETYELVEKFYDPMFKYHLTVAQVRGGMVFELANSIVLPFDCRDYAVVLRKYADKIYSISMKHPQEMKTYSVSFDSLFSAVKNFTEIASKFSERLQDFDKSNPIVLRMMNDQLMFLERAFIDPLGLPDRPFYRHVIYAPSSHNKYAGESFPGIYDALFDIESKVDPSKAWGEVKRQIYVAAFTVQAAAETLSEVA
Inhibitor
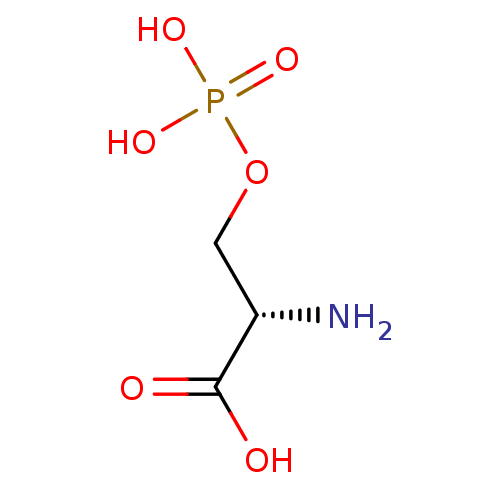
Name:
BDBM17664
Synonyms:
(2S)-2-amino-3-(phosphonooxy)propanoic acid | CHEMBL284377 | L-SOP | L-serine O-phosphate
Type:
Small organic molecule
Emp. Form.:
C3H8NO6P
Mol. Mass.:
185.0725
SMILES:
N[C@@H](COP(O)(O)=O)C(O)=O