Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM27888
Substrate
BDBM21150
Meas. Tech.
CYP3A4 Enzyme Inhibition Assay
pH
7.4±n/a
Temperature
310.15±n/a K
IC50
26000±n/a nM
Citation
 Velaparthi, U; Wittman, M; Liu, P; Carboni, JM; Lee, FY; Attar, R; Balimane, P; Clarke, W; Sinz, MW; Hurlburt, W; Patel, K; Discenza, L; Kim, S; Gottardis, M; Greer, A; Li, A; Saulnier, M; Yang, Z; Zimmermann, K; Trainor, G; Vyas, D Discovery and evaluation of 4-(2-(4-chloro-1H-pyrazol-1-yl)ethylamino)-3-(6-(1-(3-fluoropropyl)piperidin-4-yl)-4-methyl-1H-benzo[d]imidazol-2-yl)pyridin-2(1H)-one (BMS-695735), an orally efficacious inhibitor of insulin-like growth factor-1 receptor kinase with broad spectrum in vivo antitumor acti J Med Chem 51:5897-900 (2008) [PubMed] Article
Velaparthi, U; Wittman, M; Liu, P; Carboni, JM; Lee, FY; Attar, R; Balimane, P; Clarke, W; Sinz, MW; Hurlburt, W; Patel, K; Discenza, L; Kim, S; Gottardis, M; Greer, A; Li, A; Saulnier, M; Yang, Z; Zimmermann, K; Trainor, G; Vyas, D Discovery and evaluation of 4-(2-(4-chloro-1H-pyrazol-1-yl)ethylamino)-3-(6-(1-(3-fluoropropyl)piperidin-4-yl)-4-methyl-1H-benzo[d]imidazol-2-yl)pyridin-2(1H)-one (BMS-695735), an orally efficacious inhibitor of insulin-like growth factor-1 receptor kinase with broad spectrum in vivo antitumor acti J Med Chem 51:5897-900 (2008) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
Inhibitor
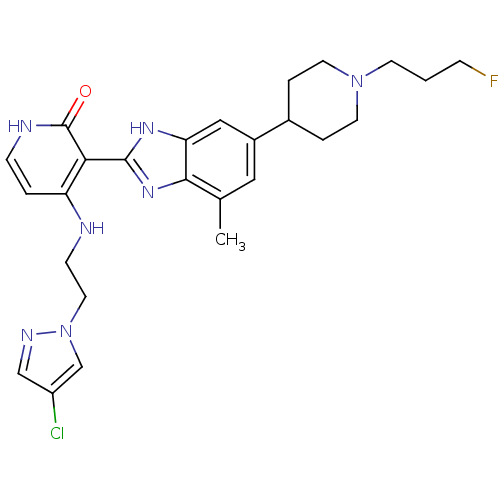
Name:
BDBM27888
Synonyms:
4-{[2-(4-chloro-1H-pyrazol-1-yl)ethyl]amino}-3-{6-[1-(3-fluoropropyl)piperidin-4-yl]-4-methyl-1H-1,3-benzodiazol-2-yl}-1,2-dihydropyridin-2-one | BMS-695735
Type:
Small organic molecule
Emp. Form.:
C26H31ClFN7O
Mol. Mass.:
512.022
SMILES:
Cc1cc(cc2[nH]c(nc12)-c1c(NCCn2cc(Cl)cn2)cc[nH]c1=O)C1CCN(CCCF)CC1