Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cathepsin D
Ligand
BDBM50008520
Substrate
n/a
Meas. Tech.
ChEBML_45328
IC50
>1000000±n/a nM
Citation
 Kleemann, HW; Heitsch, H; Henning, R; Kramer, W; Kocher, W; Lerch, U; Linz, W; Nickel, WU; Ruppert, D; Urbach, H Renin inhibitory pentols showing improved enteral bioavailability. J Med Chem 35:559-67 (1992) [PubMed] Article
Kleemann, HW; Heitsch, H; Henning, R; Kramer, W; Kocher, W; Lerch, U; Linz, W; Nickel, WU; Ruppert, D; Urbach, H Renin inhibitory pentols showing improved enteral bioavailability. J Med Chem 35:559-67 (1992) [PubMed] Article More Info.:
Target
Name:
Cathepsin D
Synonyms:
CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor
Type:
Enzyme
Mol. Mass.:
44551.72
Organism:
Homo sapiens (Human)
Description:
Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated.
Residue:
412
Sequence:
MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVPAVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIHHKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFGEATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQPGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSLMVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQAGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
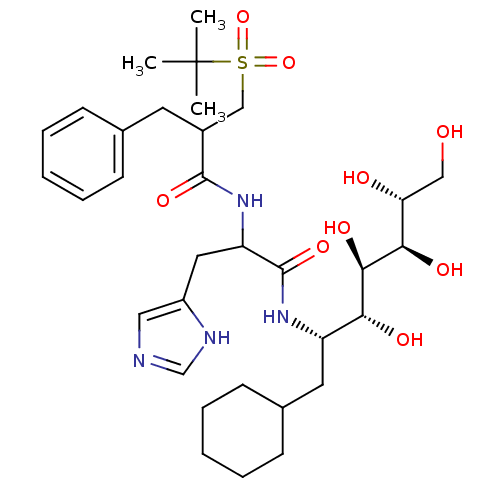
Inhibitor
Name:
BDBM50008520
Synonyms:
2-Benzyl-N-[1-(1-cyclohexylmethyl-2,3,4,5,6-pentahydroxy-hexylcarbamoyl)-2-(1H-imidazol-4-yl)-ethyl]-3-(2-methyl-propane-2-sulfonyl)-propionamide | CHEMBL357253
Type:
Small organic molecule
Emp. Form.:
C33H52N4O9S
Mol. Mass.:
680.852
SMILES:
CC(C)(C)S(=O)(=O)CC(Cc1ccccc1)C(=O)NC(Cc1cnc[nH]1)C(=O)N[C@@H](CC1CCCCC1)[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO