Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 1A
Ligand
BDBM50063277
Substrate
n/a
Meas. Tech.
ChEMBL_1451 (CHEMBL616575)
Ki
4.5±n/a nM
Citation
 Wustrow, D; Belliotti, T; Glase, S; Kesten, SR; Johnson, D; Colbry, N; Rubin, R; Blackburn, A; Akunne, H; Corbin, A; Davis, MD; Georgic, L; Whetzel, S; Zoski, K; Heffner, T; Pugsley, T; Wise, L Aminopyrimidines with high affinity for both serotonin and dopamine receptors. J Med Chem 41:760-71 (1998) [PubMed] Article
Wustrow, D; Belliotti, T; Glase, S; Kesten, SR; Johnson, D; Colbry, N; Rubin, R; Blackburn, A; Akunne, H; Corbin, A; Davis, MD; Georgic, L; Whetzel, S; Zoski, K; Heffner, T; Pugsley, T; Wise, L Aminopyrimidines with high affinity for both serotonin and dopamine receptors. J Med Chem 41:760-71 (1998) [PubMed] Article More Info.:
Target
Name:
5-hydroxytryptamine receptor 1A
Synonyms:
5-HT-1A | 5-HT1 | 5-HT1A | 5-Hydroxytryptamine receptor 1A (5-HT1A) | 5-hydroxytryptamine receptor 1A (5HT1A) | 5HT1A_RAT | 5ht1a | G-21 | Htr1a | Serotonin 1 (5-HT1) receptor | Serotonin 1a (5-HT1a) receptor/Adrenergic receptor alpha-1 | Serotonin receptor 1A
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
46445.29
Organism:
Rattus norvegicus (rat)
Description:
Binding assays were performed using rat hippocampal membranes.
Residue:
422
Sequence:
MDVFSFGQGNNTTASQEPFGTGGNVTSISDVTFSYQVITSLLLGTLIFCAVLGNACVVAAIALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCCTSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPEDRSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVRKVEKKGAGTSLGTSSAPPPKKSLNGQPGSGDWRRCAENRAVGTPCTNGAVRQGDDEATLEVIEVHRVGNSKEHLPLPSESGSNSYAPACLERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLPFFIVALVLPFCESSCHMPALLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFCRR
Inhibitor
Name:
BDBM50063277
Synonyms:
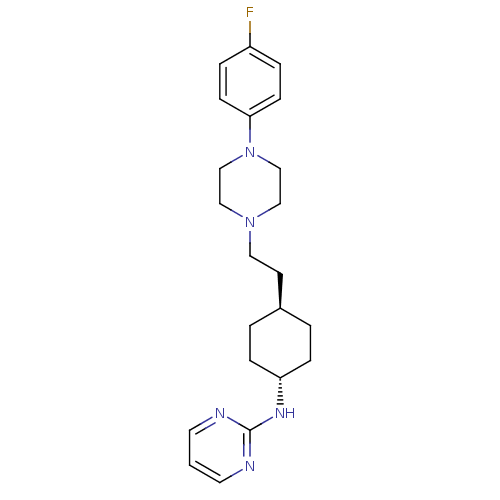
(4-{2-[4-(4-Fluoro-phenyl)-piperazin-1-yl]-ethyl}-cyclohexyl)-pyrimidin-2-yl-amine | CHEMBL349034
Type:
Small organic molecule
Emp. Form.:
C22H30FN5
Mol. Mass.:
383.5055
SMILES:
Fc1ccc(cc1)N1CCN(CC[C@H]2CC[C@@H](CC2)Nc2ncccn2)CC1 |wU:16.20,wD:13.13,(20.1,-6.75,;18.55,-6.85,;17.69,-5.56,;16.16,-5.65,;15.48,-7.01,;16.31,-8.3,;17.85,-8.23,;13.94,-7.11,;13.24,-8.49,;11.7,-8.56,;10.85,-7.27,;9.31,-7.34,;8.46,-6.05,;6.93,-6.12,;6.09,-4.83,;4.55,-4.9,;3.84,-6.29,;4.69,-7.57,;6.23,-7.5,;2.3,-6.36,;1.45,-5.04,;2.18,-3.69,;1.34,-2.37,;-.21,-2.44,;-.91,-3.83,;-.07,-5.14,;11.56,-5.89,;13.09,-5.82,)|