Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Chymase
Ligand
BDBM50068901
Substrate
n/a
Meas. Tech.
ChEBML_49442
Ki
1.3±n/a nM
Citation
 Eda, M; Ashimori, A; Akahoshi, F; Yoshimura, T; Inoue, Y; Fukaya, C; Nakajima, M; Fukuyama, H; Imada, T; Takai, S; Shiota, N; Miyazaki, M; Nakamura, N Peptidyl human heart chymase inhibitors. 1. Synthesis and inhibitory activity of difluoromethylene ketone derivatives bearing P' binding subsites. Bioorg Med Chem Lett 8:913-8 (1999) [PubMed] Article
Eda, M; Ashimori, A; Akahoshi, F; Yoshimura, T; Inoue, Y; Fukaya, C; Nakajima, M; Fukuyama, H; Imada, T; Takai, S; Shiota, N; Miyazaki, M; Nakamura, N Peptidyl human heart chymase inhibitors. 1. Synthesis and inhibitory activity of difluoromethylene ketone derivatives bearing P' binding subsites. Bioorg Med Chem Lett 8:913-8 (1999) [PubMed] Article More Info.:
Target
Name:
Chymase
Synonyms:
Alpha-chymase | CMA1 | CMA1_HUMAN | CYH | CYM | Chymase precursor | Mast cell protease I
Type:
Enzyme
Mol. Mass.:
27340.12
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
247
Sequence:
MLLLPLPLLLFLLCSRAEAGEIIGGTECKPHSRPYMAYLEIVTSNGPSKFCGGFLIRRNFVLTAAHCAGRSITVTLGAHNITEEEDTWQKLEVIKQFRHPKYNTSTLHHDIMLLKLKEKASLTLAVGTLPFPSQFNFVPPGRMCRVAGWGRTGVLKPGSDTLQEVKLRLMDPQACSHFRDFDHNLQLCVGNPRKTKSAFKGDSGGPLLCAGVAQGIVSYGRSDAKPPAVFTRISHYRPWINQILQAN
Inhibitor
Name:
BDBM50068901
Synonyms:
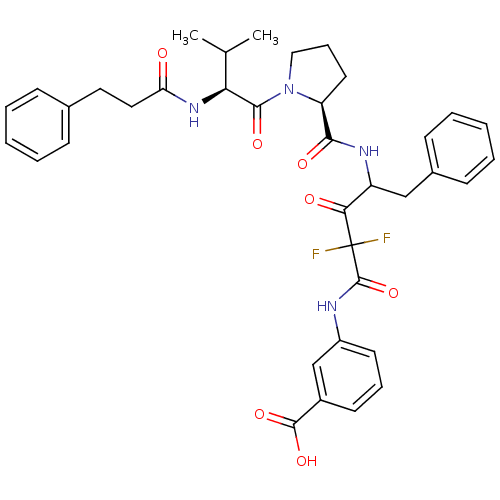
3-[2,2-Difluoro-4-({(S)-1-[(S)-3-methyl-2-(3-phenyl-propionylamino)-butyryl]-pyrrolidine-2-carbonyl}-amino)-3-oxo-5-phenyl-pentanoylamino]-benzoic acid | CHEMBL353816
Type:
Small organic molecule
Emp. Form.:
C37H40F2N4O7
Mol. Mass.:
690.7329
SMILES:
CC(C)[C@H](NC(=O)CCc1ccccc1)C(=O)N1CCC[C@H]1C(=O)NC(Cc1ccccc1)C(=O)C(F)(F)C(=O)Nc1cccc(c1)C(O)=O