Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
72 kDa type IV collagenase
Ligand
BDBM50120691
Substrate
n/a
Meas. Tech.
ChEMBL_104397 (CHEMBL715000)
Ki
<2.8±n/a nM
Citation
 Duan, JJ; Chen, L; Wasserman, ZR; Lu, Z; Liu, RQ; Covington, MB; Qian, M; Hardman, KD; Magolda, RL; Newton, RC; Christ, DD; Wexler, RR; Decicco, CP Discovery of gamma-lactam hydroxamic acids as selective inhibitors of tumor necrosis factor alpha converting enzyme: design, synthesis, and structure-activity relationships. J Med Chem 45:4954-7 (2002) [PubMed] Article
Duan, JJ; Chen, L; Wasserman, ZR; Lu, Z; Liu, RQ; Covington, MB; Qian, M; Hardman, KD; Magolda, RL; Newton, RC; Christ, DD; Wexler, RR; Decicco, CP Discovery of gamma-lactam hydroxamic acids as selective inhibitors of tumor necrosis factor alpha converting enzyme: design, synthesis, and structure-activity relationships. J Med Chem 45:4954-7 (2002) [PubMed] Article More Info.:
Target
Name:
72 kDa type IV collagenase
Synonyms:
72 kDa gelatinase | 72 kDa type IV collagenase precursor | CLG4A | Gelatinase A | Gelatinase A (MMP-2) | MMP2 | MMP2_HUMAN | Matrix metalloproteinase-2 | Matrix metalloproteinase-2 (MMP 2) | Matrix metalloproteinase-2 (MMP2) | Matrix metalloproteinases 2 (MMP-2) | TBE-1
Type:
Enzyme
Mol. Mass.:
73870.36
Organism:
Homo sapiens (Human)
Description:
P08253
Residue:
660
Sequence:
MEALMARGALTGPLRALCLLGCLLSHAAAAPSPIIKFPGDVAPKTDKELAVQYLNTFYGCPKESCNLFVLKDTLKKMQKFFGLPQTGDLDQNTIETMRKPRCGNPDVANYNFFPRKPKWDKNQITYRIIGYTPDLDPETVDDAFARAFQVWSDVTPLRFSRIHDGEADIMINFGRWEHGDGYPFDGKDGLLAHAFAPGTGVGGDSHFDDDELWTLGEGQVVRVKYGNADGEYCKFPFLFNGKEYNSCTDTGRSDGFLWCSTTYNFEKDGKYGFCPHEALFTMGGNAEGQPCKFPFRFQGTSYDSCTTEGRTDGYRWCGTTEDYDRDKKYGFCPETAMSTVGGNSEGAPCVFPFTFLGNKYESCTSAGRSDGKMWCATTANYDDDRKWGFCPDQGYSLFLVAAHEFGHAMGLEHSQDPGALMAPIYTYTKNFRLSQDDIKGIQELYGASPDIDLGTGPTPTLGPVTPEICKQDIVFDGIAQIRGEIFFFKDRFIWRTVTPRDKPMGPLLVATFWPELPEKIDAVYEAPQEEKAVFFAGNEYWIYSASTLERGYPKPLTSLGLPPDVQRVDAAFNWSKNKKTYIFAGDKFWRYNEVKKKMDPGFPKLIADAWNAIPDNLDAVVDLQGGGHSYFFKGAYYLKLENQSLKSVKFGSIKSDWLGC
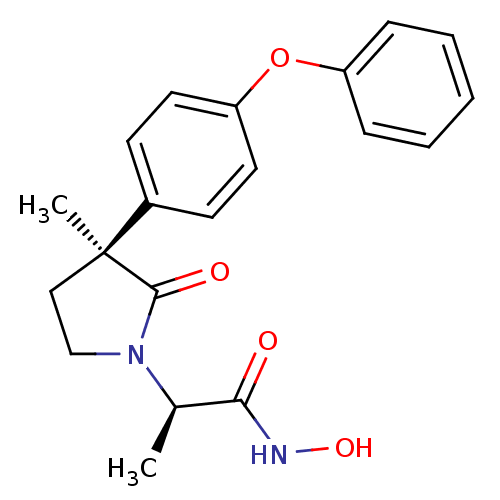
Inhibitor
Name:
BDBM50120691
Synonyms:
(R)-N-hydroxy-2-((S)-3-methyl-2-oxo-3-(4-phenoxyphenyl)pyrrolidin-1-yl)propanamide | CHEMBL144049 | N-Hydroxy-2-[3-methyl-2-oxo-3-(4-phenoxy-phenyl)-pyrrolidin-1-yl]-propionamide
Type:
Small organic molecule
Emp. Form.:
C20H22N2O4
Mol. Mass.:
354.3997
SMILES:
C[C@@H](N1CC[C@](C)(C1=O)c1ccc(Oc2ccccc2)cc1)C(=O)NO |r|