Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Sodium/hydrogen exchanger 2
Ligand
BDBM50058759
Substrate
n/a
Meas. Tech.
ChEMBL_397781 (CHEMBL856312)
IC50
62000±n/a nM
Citation
 Atwal, KS; O'Neil, SV; Ahmad, S; Doweyko, L; Kirby, M; Dorso, CR; Chandrasena, G; Chen, BC; Zhao, R; Zahler, R Synthesis and biological activity of 5-aryl-4-(4-(5-methyl-1H-imidazol-4-yl)piperidin-1-yl)pyrimidine analogs as potent, highly selective, and orally bioavailable NHE-1 inhibitors. Bioorg Med Chem Lett 16:4796-9 (2006) [PubMed] Article
Atwal, KS; O'Neil, SV; Ahmad, S; Doweyko, L; Kirby, M; Dorso, CR; Chandrasena, G; Chen, BC; Zhao, R; Zahler, R Synthesis and biological activity of 5-aryl-4-(4-(5-methyl-1H-imidazol-4-yl)piperidin-1-yl)pyrimidine analogs as potent, highly selective, and orally bioavailable NHE-1 inhibitors. Bioorg Med Chem Lett 16:4796-9 (2006) [PubMed] Article More Info.:
Target
Name:
Sodium/hydrogen exchanger 2
Synonyms:
NHE-2 | NHE2 | Na(+)/H(+) exchanger 2 | SL9A2_HUMAN | SLC9A2 | Solute carrier family 9 member 2
Type:
PROTEIN
Mol. Mass.:
91542.38
Organism:
Homo sapiens (Human)
Description:
ChEMBL_397781
Residue:
812
Sequence:
MEPLGNWRSLRAPLPPMLLLLLLQVAGPVGALAETLLNAPRAMGTSSSPPSPASVVAPGTTLFEESRLPVFTLDYPHVQIPFEITLWILLASLAKIGFHLYHKLPTIVPESCLLIMVGLLLGGIIFGVDEKSPPAMKTDVFFLYLLPPIVLDAGYFMPTRPFFENIGTIFWYAVVGTLWNSIGIGVSLFGICQIEAFGLSDITLLQNLLFGSLISAVDPVAVLAVFENIHVNEQLYILVFGESLLNDAVTVVLYNLFKSFCQMKTIETIDVFAGIANFFVVGIGGVLIGIFLGFIAAFTTRFTHNIRVIEPLFVFLYSYLSYITAEMFHLSGIMAITACAMTMNKYVEENVSQKSYTTIKYFMKMLSSVSETLIFIFMGVSTVGKNHEWNWAFVCFTLAFCLMWRALGVFVLTQVINRFRTIPLTFKDQFIIAYGGLRGAICFALVFLLPAAVFPRKKLFITAAIVVIFFTVFILGITIRPLVEFLDVKRSNKKQQAVSEEIYCRLFDHVKTGIEDVCGHWGHNFWRDKFKKFDDKYLRKLLIRENQPKSSIVSLYKKLEIKHAIEMAETGMISTVPTFASLNDCREEKIRKVTSSETDEIRELLSRNLYQIRQRTLSYNRHSLTADTSERQAKEILIRRRHSLRESIRKDSSLNREHRASTSTSRYLSLPKNTKLPEKLQKRRTISIADGNSSDSDADAGTTVLNLQPRARRFLPEQFSKKSPQSYKMEWKNEVDVDSGRDMPSTPPTPHSREKGTQTSGLLQQPLLSKDQSGSEREDSLTEGIPPKPPPRLVWRASEPGSRKARFGSEKP
Inhibitor
Name:
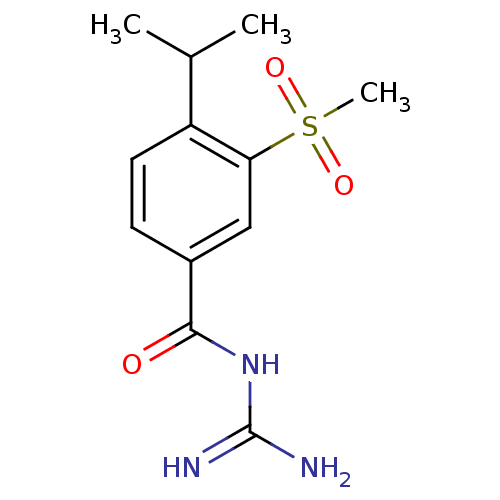
BDBM50058759
Synonyms:
CHEMBL436559 | CHEMBL462831 | Cariporide | N-(4-Isopropyl-3-methanesulfonyl-benzoyl)-guanidine (Cariporide) | N-(4-Isopropyl-3-methanesulfonyl-benzoyl)-guanidine(Cariporide) | N-(4-isopropyl-3-methanesulfonyl-benzoyl)-guanidine | N-(diaminomethylene)-4-isopropyl-3-(methylsulfonyl)benzamide
Type:
Small organic molecule
Emp. Form.:
C12H17N3O3S
Mol. Mass.:
283.347
SMILES:
CC(C)c1ccc(cc1S(C)(=O)=O)C(=O)NC(N)=N