Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 2A
Ligand
BDBM50130293
Substrate
n/a
Meas. Tech.
ChEMBL_558364 (CHEMBL954166)
Ki
8.7±n/a nM
Citation
 Butini, S; Gemma, S; Campiani, G; Franceschini, S; Trotta, F; Borriello, M; Ceres, N; Ros, S; Coccone, SS; Bernetti, M; De Angelis, M; Brindisi, M; Nacci, V; Fiorini, I; Novellino, E; Cagnotto, A; Mennini, T; Sandager-Nielsen, K; Andreasen, JT; Scheel-Kruger, J; Mikkelsen, JD; Fattorusso, C Discovery of a new class of potential multifunctional atypical antipsychotic agents targeting dopamine D3 and serotonin 5-HT1A and 5-HT2A receptors: design, synthesis, and effects on behavior. J Med Chem 52:151-69 (2009) [PubMed] Article
Butini, S; Gemma, S; Campiani, G; Franceschini, S; Trotta, F; Borriello, M; Ceres, N; Ros, S; Coccone, SS; Bernetti, M; De Angelis, M; Brindisi, M; Nacci, V; Fiorini, I; Novellino, E; Cagnotto, A; Mennini, T; Sandager-Nielsen, K; Andreasen, JT; Scheel-Kruger, J; Mikkelsen, JD; Fattorusso, C Discovery of a new class of potential multifunctional atypical antipsychotic agents targeting dopamine D3 and serotonin 5-HT1A and 5-HT2A receptors: design, synthesis, and effects on behavior. J Med Chem 52:151-69 (2009) [PubMed] Article More Info.:
Target
Name:
5-hydroxytryptamine receptor 2A
Synonyms:
5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
52852.05
Organism:
Rattus norvegicus (rat)
Description:
Rat cortex membranes 5-HT2A receptors.
Residue:
471
Sequence:
MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGYLPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIADMLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNPIHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSFVAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIHREPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGALLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYKSSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
Inhibitor
Name:
BDBM50130293
Synonyms:
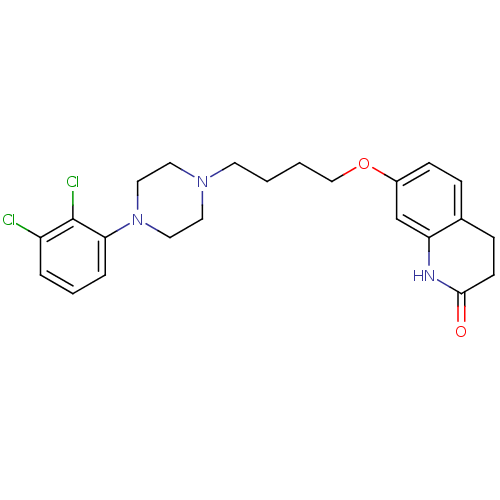
7-{4-[4-(2,3-dichlorophenyl)piperazin-1-yl]butoxy}-3,4-dihydroquinolin-2(1H)-one | ARIPIPRAZOLE | CHEMBL1112 | US10174011, aripiprazole | US10752588, Compound Aripiprazole | US11498896, Compound Aripiprazole | US9018213, aripprazole | US9156822, Aripiprazole
Type:
Small organic molecule
Emp. Form.:
C23H27Cl2N3O2
Mol. Mass.:
448.385
SMILES:
Clc1cccc(N2CCN(CCCCOc3ccc4CCC(=O)Nc4c3)CC2)c1Cl