Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50337730
Substrate
n/a
Meas. Tech.
ChEMBL_726744 (CHEMBL1686218)
IC50
1.4±n/a nM
Citation
 De Savi, C; Pape, A; Cumming, JG; Ting, A; Smith, PD; Burrows, JN; Mills, M; Davies, C; Lamont, S; Milne, D; Cook, C; Moore, P; Sawyer, Y; Gerhardt, S The design and synthesis of novel N-hydroxyformamide inhibitors of ADAM-TS4 for the treatment of osteoarthritis. Bioorg Med Chem Lett 21:1376-81 (2011) [PubMed] Article
De Savi, C; Pape, A; Cumming, JG; Ting, A; Smith, PD; Burrows, JN; Mills, M; Davies, C; Lamont, S; Milne, D; Cook, C; Moore, P; Sawyer, Y; Gerhardt, S The design and synthesis of novel N-hydroxyformamide inhibitors of ADAM-TS4 for the treatment of osteoarthritis. Bioorg Med Chem Lett 21:1376-81 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
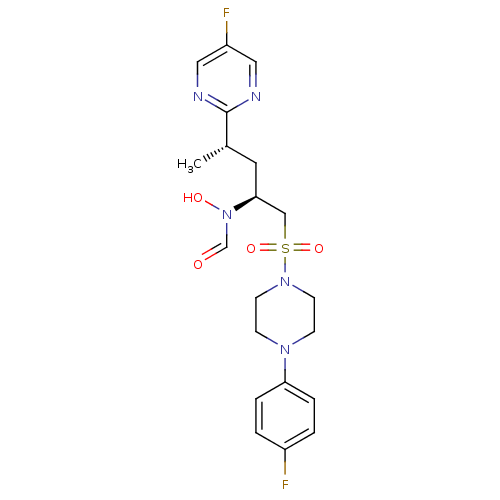
Inhibitor
Name:
BDBM50337730
Synonyms:
CHEMBL1683443 | N-((2S,4S)-1-(4-(4-fluorophenyl)piperazin-1-ylsulfonyl)-4-(5-fluoropyrimidin-2-yl)pentan-2-yl)-N-hydroxyformamide
Type:
Small organic molecule
Emp. Form.:
C20H25F2N5O4S
Mol. Mass.:
469.505
SMILES:
C[C@@H](C[C@@H](CS(=O)(=O)N1CCN(CC1)c1ccc(F)cc1)N(O)C=O)c1ncc(F)cn1 |r|