Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cholecystokinin receptor type A
Ligand
BDBM50019241
Substrate
n/a
Meas. Tech.
ChEBML_49558
IC50
71±n/a nM
Citation
 Evans, BE; Rittle, KE; Bock, MG; DiPardo, RM; Freidinger, RM; Whitter, WL; Lundell, GF; Veber, DF; Anderson, PS; Chang, RS Methods for drug discovery: development of potent, selective, orally effective cholecystokinin antagonists. J Med Chem 31:2235-46 (1989) [PubMed] Article
Evans, BE; Rittle, KE; Bock, MG; DiPardo, RM; Freidinger, RM; Whitter, WL; Lundell, GF; Veber, DF; Anderson, PS; Chang, RS Methods for drug discovery: development of potent, selective, orally effective cholecystokinin antagonists. J Med Chem 31:2235-46 (1989) [PubMed] Article More Info.:
Target
Name:
Cholecystokinin receptor type A
Synonyms:
CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A
Type:
Enzyme Catalytic Domain
Mol. Mass.:
49676.37
Organism:
RAT
Description:
Cholecystokinin central 0 RAT::P30551
Residue:
444
Sequence:
MSHSPARQHLVESSRMDVVDSLLMNGSNITPPCELGLENETLFCLDQPQPSKEWQSALQILLYSIIFLLSVLGNTLVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLKDFIFGSAVCKTTTYFMGTSVSVSTFNLVAISLERYGAICRPLQSRVWQTKSHALKVIAATWCLSFTIMTPYPIYSNLVPFTKNNNQTANMCRFLLPSDAMQQSWQTFLLLILFLLPGIVMVVAYGLISLELYQGIKFDASQKKSAKEKKPSTGSSTRYEDSDGCYLQKSRPPRKLELQQLSSGSGGSRLNRIRSSSSAANLIAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTVSAEKHLSGTPISFILLLSYTSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGVRGEVGEEEDGRTIRALLSRYSYSHMSTSAPPP
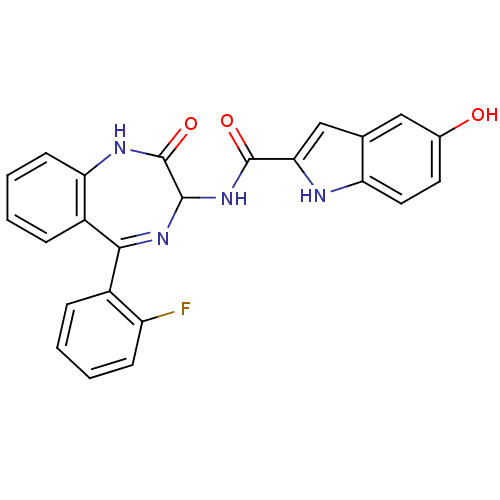
Inhibitor
Name:
BDBM50019241
Synonyms:
5-Hydroxy-1H-indole-2-carboxylic acid [5-(2-fluoro-phenyl)-2-oxo-2,3-dihydro-1H-benzo[e][1,4]diazepin-3-yl]-amide | 5-Hydroxy-1H-indole-2-carboxylic acid [5-(2-fluoro-phenyl)-2-oxo-2,3-dihydro-1H-benzo[e][1,4]diazepin-3-yl]-amide: : 0.2CHCl3 | CHEMBL74066
Type:
Small organic molecule
Emp. Form.:
C24H17FN4O3
Mol. Mass.:
428.4152
SMILES:
Oc1ccc2[nH]c(cc2c1)C(=O)NC1N=C(c2ccccc2F)c2ccccc2NC1=O |t:16|