Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Stromelysin-1
Ligand
BDBM50102631
Substrate
n/a
Meas. Tech.
ChEMBL_104875 (CHEMBL709173)
Ki
2.9±n/a nM
Citation
 Xue, CB; Voss, ME; Nelson, DJ; Duan, JJ; Cherney, RJ; Jacobson, IC; He, X; Roderick, J; Chen, L; Corbett, RL; Wang, L; Meyer, DT; Kennedy, K; DeGradodagger, WF; Hardman, KD; Teleha, CA; Jaffee, BD; Liu, RQ; Copeland, RA; Covington, MB; Christ, DD; Trzaskos, JM; Newton, RC; Magolda, RL; Wexler, RR; Decicco, CP Design, synthesis, and structure-activity relationships of macrocyclic hydroxamic acids that inhibit tumor necrosis factor alpha release in vitro and in vivo. J Med Chem 44:2636-60 (2001) [PubMed] Article
Xue, CB; Voss, ME; Nelson, DJ; Duan, JJ; Cherney, RJ; Jacobson, IC; He, X; Roderick, J; Chen, L; Corbett, RL; Wang, L; Meyer, DT; Kennedy, K; DeGradodagger, WF; Hardman, KD; Teleha, CA; Jaffee, BD; Liu, RQ; Copeland, RA; Covington, MB; Christ, DD; Trzaskos, JM; Newton, RC; Magolda, RL; Wexler, RR; Decicco, CP Design, synthesis, and structure-activity relationships of macrocyclic hydroxamic acids that inhibit tumor necrosis factor alpha release in vitro and in vivo. J Med Chem 44:2636-60 (2001) [PubMed] Article More Info.:
Target
Name:
Stromelysin-1
Synonyms:
MMP-3 | MMP3 | MMP3_HUMAN | Matrix metalloproteinase (2 and 3) | Matrix metalloproteinase 3 | Matrix metalloproteinase-3 | Matrix metalloproteinase-3 (MMP-3) | Matrix metalloproteinase-3 (MMP3) | SL-1 | STMY1 | Stromelysin 1 | Transin-1
Type:
Enzyme
Mol. Mass.:
53973.13
Organism:
Homo sapiens (Human)
Description:
P08254
Residue:
477
Sequence:
MKSLPILLLLCVAVCSAYPLDGAARGEDTSMNLVQKYLENYYDLKKDVKQFVRRKDSGPVVKKIREMQKFLGLEVTGKLDSDTLEVMRKPRCGVPDVGHFRTFPGIPKWRKTHLTYRIVNYTPDLPKDAVDSAVEKALKVWEEVTPLTFSRLYEGEADIMISFAVREHGDFYPFDGPGNVLAHAYAPGPGINGDAHFDDDEQWTKDTTGTNLFLVAAHEIGHSLGLFHSANTEALMYPLYHSLTDLTRFRLSQDDINGIQSLYGPPPDSPETPLVPTEPVPPEPGTPANCDPALSFDAVSTLRGEILIFKDRHFWRKSLRKLEPELHLISSFWPSLPSGVDAAYEVTSKDLVFIFKGNQFWAIRGNEVRAGYPRGIHTLGFPPTVRKIDAAISDKEKNKTYFFVEDKYWRFDEKRNSMEPGFPKQIAEDFPGIDSKIDAVFEEFGFFYFFTGSSQLEFDPNAKKVTHTLKSNSWLNC
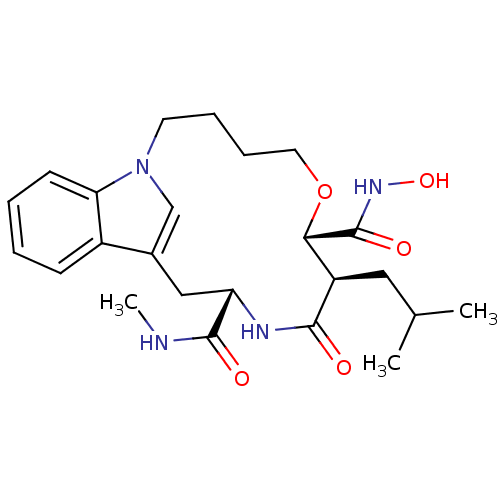
Inhibitor
Name:
BDBM50102631
Synonyms:
8-Isobutyl-9-oxo-6-oxa-1,10-diaza-tricyclo[11.6.1.0*14,19*]icosa-13(20),14(19),15,17-tetraene-7,11-dicarboxylic acid 7-hydroxyamide 11-methylamide | CHEMBL88718
Type:
Small organic molecule
Emp. Form.:
C24H34N4O5
Mol. Mass.:
458.5506
SMILES:
CNC(=O)[C@@H]1Cc2cn(CCCCO[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)c1ccccc21