Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Phosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta
Ligand
BDBM12915
Substrate
n/a
Meas. Tech.
ChEMBL_438490 (CHEMBL887590)
IC50
2100±n/a nM
Citation
 Hayakawa, M; Kaizawa, H; Moritomo, H; Koizumi, T; Ohishi, T; Yamano, M; Okada, M; Ohta, M; Tsukamoto, S; Raynaud, FI; Workman, P; Waterfield, MD; Parker, P Synthesis and biological evaluation of pyrido[3',2':4,5]furo[3,2-d]pyrimidine derivatives as novel PI3 kinase p110alpha inhibitors. Bioorg Med Chem Lett 17:2438-42 (2007) [PubMed] Article
Hayakawa, M; Kaizawa, H; Moritomo, H; Koizumi, T; Ohishi, T; Yamano, M; Okada, M; Ohta, M; Tsukamoto, S; Raynaud, FI; Workman, P; Waterfield, MD; Parker, P Synthesis and biological evaluation of pyrido[3',2':4,5]furo[3,2-d]pyrimidine derivatives as novel PI3 kinase p110alpha inhibitors. Bioorg Med Chem Lett 17:2438-42 (2007) [PubMed] Article More Info.:
Target
Name:
Phosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit beta
Synonyms:
C2-PI3K | P3C2B_HUMAN | PI3K-C2beta | PIK3C2B | Phosphatidylinositol 4-phosphate 3-kinase C2 beta (PIK3C2B) | Phosphatidylinositol-4-phosphate 3-kinase C2 domain-containing beta polypeptide | Phosphoinositide 3-Kinase (PI3K), C2beta | Phosphoinositide 3-Kinase-C2-beta | PtdIns-3-kinase C2 beta
Type:
Enzyme
Mol. Mass.:
184784.86
Organism:
Homo sapiens (Human)
Description:
O00750
Residue:
1634
Sequence:
MSSTQGNGEHWKSLESVGISRKELAMAEALQMEYDALSRLRHDKEENRAKQNADPSLISWDEPGVDFYSKPAGRRTDLKLLRGLSGSDPTLNYNSLSPQEGPPNHSTSQGPQPGSDPWPKGSLSGDYLYIFDGSDGGVSSSPGPGDIEGSCKKLSPPPLPPRASIWDTPPLPPRKGSPSSSKISQPSDINTFSLVEQLPGKLLEHRILEEEEVLGGGGQGRLLGSVDYDGINDAITRLNLKSTYDAEMLRDATRGWKEGRGPLDFSKDTSGKPVARSKTMPPQVPPRTYASRYGNRKNATPGKNRRISAAPVGSRPHTVANGHELFEVSEERDEEVAAFCHMLDILRSGSDIQDYFLTGYVWSAVTPSPEHLGDEVNLKVTVLCDRLQEALTFTCNCSSTVDLLIYQTLCYTHDDLRNVDVGDFVLKPCGLEEFLQNKHALGSHEYIQYCRKFDIDIRLQLMEQKVVRSDLARTVNDDQSPSTLNYLVHLQERPVKQTISRQALSLLFDTYHNEVDAFLLADGDFPLKADRVVQSVKAICNALAAVETPEITSALNQLPPCPSRMQPKIQKDPSVLAVRENREKVVEALTAAILDLVELYCNTFNADFQTAVPGSRKHDLVQEACHFARSLAFTVYATHRIPIIWATSYEDFYLSCSLSHGGKELCSPLQTRRAHFSKYLFHLIVWDQQICFPVQVNRLPRETLLCATLYALPIPPPGSSSEANKQRRVPEALGWVTTPLFNFRQVLTCGRKLLGLWPATQENPSARWSAPNFHQPDSVILQIDFPTSAFDIKFTSPPGDKFSPRYEFGSLREEDQRKLKDIMQKESLYWLTDADKKRLWEKRYYCHSEVSSLPLVLASAPSWEWACLPDIYVLLKQWTHMNHQDALGLLHATFPDQEVRRMAVQWIGSLSDAELLDYLPQLVQALKYECYLDSPLVRFLLKRAVSDLRVTHYFFWLLKDGLKDSQFSIRYQYLLAALLCCCGKGLREEFNRQCWLVNALAKLAQQVREAAPSARQGILRTGLEEVKQFFALNGSCRLPLSPSLLVKGIVPRDCSYFNSNAVPLKLSFQNVDPLGENIRVIFKCGDDLRQDMLTLQMIRIMSKIWVQEGLDMRMVIFRCFSTGRGRGMVEMIPNAETLRKIQVEHGVTGSFKDRPLADWLQKHNPGEDEYEKAVENFIYSCAGCCVATYVLGICDRHNDNIMLKTTGHMFHIDFGRFLGHAQMFGNIKRDRAPFVFTSDMAYVINGGDKPSSRFHDFVDLCCQAYNLIRKHTHLFLNLLGLMLSCGIPELSDLEDLKYVYDALRPQDTEANATTYFTRLIESSLGSVATKLNFFIHNLAQMKFTGSDDRLTLSFASRTHTLKSSGRISDVFLCRHEKIFHPNKGYIYVVKVMRENTHEATYIQRTFEEFQELHNKLRLLFPSSHLPSFPSRFVIGRSRGEAVAERRREELNGYIWHLIHAPPEVAECDLVYTFFHPLPRDEKAMGTSPAPKSSDGTWARPVGKVGGEVKLSISYKNNKLFIMVMHIRGLQLLQDGNDPDPYVKIYLLPDPQKTTKRKTKVARKTCNPTYNEMLVYDGIPKGDLQQRELQLSVLSEQGFWENVLLGEVNIRLRELDLAQEKTGWFALGSRSHGTL
Inhibitor
Name:
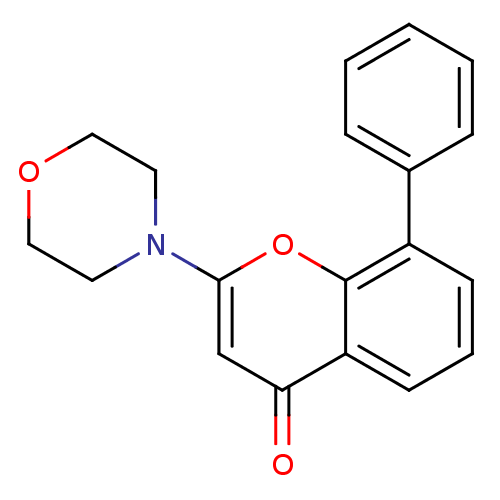
BDBM12915
Synonyms:
2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one | 2-(morpholin-4-yl)-8-phenyl-4H-chromen-4-one | CHEMBL98350 | LY294002 | US10308662, Compound LY294002 | US9505780, LY294002
Type:
Small organic molecule
Emp. Form.:
C19H17NO3
Mol. Mass.:
307.3432
SMILES:
O=c1cc(oc2c(cccc12)-c1ccccc1)N1CCOCC1