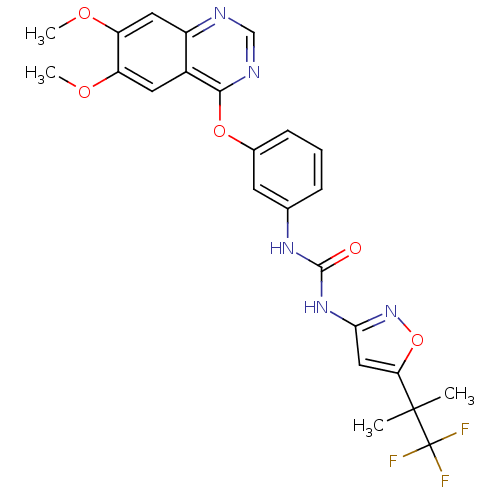
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 1A2
Ligand
BDBM50382959
Substrate
n/a
Meas. Tech.
ChEMBL_818037 (CHEMBL2033565)
IC50
>10000±n/a nM
Citation
 Rowbottom, MW; Faraoni, R; Chao, Q; Campbell, BT; Lai, AG; Setti, E; Ezawa, M; Sprankle, KG; Abraham, S; Tran, L; Struss, B; Gibney, M; Armstrong, RC; Gunawardane, RN; Nepomuceno, RR; Valenta, I; Hua, H; Gardner, MF; Cramer, MD; Gitnick, D; Insko, DE; Apuy, JL; Jones-Bolin, S; Ghose, AK; Herbertz, T; Ator, MA; Dorsey, BD; Ruggeri, B; Williams, M; Bhagwat, S; James, J; Holladay, MW Identification of 1-(3-(6,7-dimethoxyquinazolin-4-yloxy)phenyl)-3-(5-(1,1,1-trifluoro-2-methylpropan-2-yl)isoxazol-3-yl)urea hydrochloride (CEP-32496), a highly potent and orally efficacious inhibitor of V-RAF murine sarcoma viral oncogene homologue B1 (BRAF) V600E. J Med Chem 55:1082-105 (2012) [PubMed] Article
Rowbottom, MW; Faraoni, R; Chao, Q; Campbell, BT; Lai, AG; Setti, E; Ezawa, M; Sprankle, KG; Abraham, S; Tran, L; Struss, B; Gibney, M; Armstrong, RC; Gunawardane, RN; Nepomuceno, RR; Valenta, I; Hua, H; Gardner, MF; Cramer, MD; Gitnick, D; Insko, DE; Apuy, JL; Jones-Bolin, S; Ghose, AK; Herbertz, T; Ator, MA; Dorsey, BD; Ruggeri, B; Williams, M; Bhagwat, S; James, J; Holladay, MW Identification of 1-(3-(6,7-dimethoxyquinazolin-4-yloxy)phenyl)-3-(5-(1,1,1-trifluoro-2-methylpropan-2-yl)isoxazol-3-yl)urea hydrochloride (CEP-32496), a highly potent and orally efficacious inhibitor of V-RAF murine sarcoma viral oncogene homologue B1 (BRAF) V600E. J Med Chem 55:1082-105 (2012) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 1A2
Synonyms:
CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3
Type:
Enzyme
Mol. Mass.:
58423.38
Organism:
Homo sapiens (Human)
Description:
P05177
Residue:
516
Sequence:
MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKNPHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDGQSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELMAGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFPILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGNLIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLSDRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPELWEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLEFSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN