Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Apelin receptor
Ligand
BDBM499522
Substrate
n/a
Meas. Tech.
[35S]GTPgammaS Binding
EC50
1.000±n/a nM
Citation
 Chen, Y; Dransfield, PJ; Harvey, JS; Heath, JA; Houze, J; Khakoo, AY; Kopecky, DJ; Lai, S; Ma, Z; Nishimura, N; Pattaropong, V; Swaminath, G; Yeh, W; Ramsden, PD Cycloalkyl substituted triazole compounds as agonists of the APJ receptor US Patent US11020395 Publication Date 6/1/2021
Chen, Y; Dransfield, PJ; Harvey, JS; Heath, JA; Houze, J; Khakoo, AY; Kopecky, DJ; Lai, S; Ma, Z; Nishimura, N; Pattaropong, V; Swaminath, G; Yeh, W; Ramsden, PD Cycloalkyl substituted triazole compounds as agonists of the APJ receptor US Patent US11020395 Publication Date 6/1/2021 More Info.:
Target
Name:
Apelin receptor
Synonyms:
AGTRL1 | APJ | APJ_HUMAN | APLNR | Angiotensin receptor-like 1 | Apelin receptor | Apelin receptor (APJ) | G-protein coupled receptor APJ | G-protein coupled receptor HG11
Type:
Enzyme Catalytic Domain
Mol. Mass.:
42664.06
Organism:
Homo sapiens (Human)
Description:
P35414
Residue:
380
Sequence:
MEEGGDFDNYYGADNQSECEYTDWKSSGALIPAIYMLVFLLGTTGNGLVLWTVFRSSREKRRSADIFIASLAVADLTFVVTLPLWATYTYRDYDWPFGTFFCKLSSYLIFVNMYASVFCLTGLSFDRYLAIVRPVANARLRLRVSGAVATAVLWVLAALLAMPVMVLRTTGDLENTTKVQCYMDYSMVATVSSEWAWEVGLGVSSTTVGFVVPFTIMLTCYFFIAQTIAGHFRKERIEGLRKRRRLLSIIVVLVVTFALCWMPYHLVKTLYMLGSLLHWPCDFDLFLMNIFPYCTCISYVNSCLNPFLYAFFDPRFRQACTSMLCCGQSRCAGTSHSSSGEKSASYSSGHSQGPGPNMGKGGEQMHEKSIPYSQETLVVD
Inhibitor
Name:
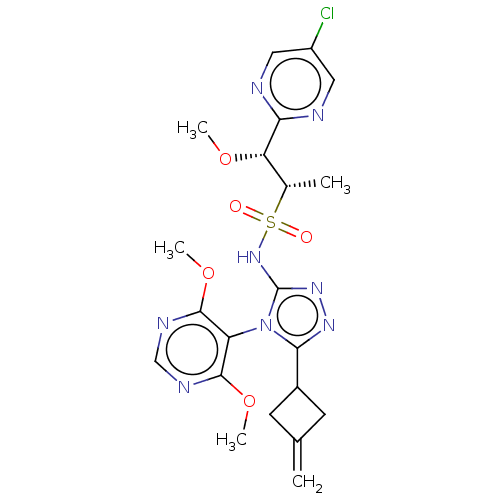
BDBM499522
Synonyms:
US11020395, Example 118.0
Type:
Small organic molecule
Emp. Form.:
C21H25ClN8O5S
Mol. Mass.:
536.992
SMILES:
CO[C@@H]([C@H](C)S(=O)(=O)Nc1nnc(C2CC(=C)C2)n1-c1c(OC)ncnc1OC)c1ncc(Cl)cn1 |r,wU:2.1,3.3,(5.21,-3.92,;3.87,-3.15,;3.87,-1.61,;2.54,-.84,;2.54,.7,;1.21,-1.61,;-.13,-2.38,;2.54,-2.38,;-.13,-.84,;-1.46,-1.61,;-1.94,-3.07,;-3.48,-3.07,;-3.95,-1.61,;-5.49,-1.61,;-6.58,-.52,;-7.67,-1.61,;-9.21,-1.61,;-6.58,-2.7,;-2.71,-.7,;-2.71,.84,;-4.04,1.61,;-5.37,.84,;-6.71,1.61,;-4.04,3.15,;-2.71,3.92,;-1.37,3.15,;-1.37,1.61,;-.04,.84,;1.3,1.61,;5.21,-.84,;6.54,-1.61,;7.88,-.84,;7.88,.7,;9.21,1.47,;6.54,1.47,;5.21,.7,)|