Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Plasmepsin I
Ligand
BDBM7991
Substrate
Peptide Substrate
Meas. Tech.
Enzyme Inhibition Measurements
Ki
24±n/a nM
Citation
 Johansson, PO; Chen, Y; Belfrage, AK; Blackman, MJ; Kvarnstrom, I; Jansson, K; Vrang, L; Hamelink, E; Hallberg, A; Rosenquist, A; Samuelsson, B Design and synthesis of potent inhibitors of the malaria aspartyl proteases plasmepsin I and II. Use of solid-phase synthesis to explore novel statine motifs. J Med Chem 47:3353-66 (2004) [PubMed] Article
Johansson, PO; Chen, Y; Belfrage, AK; Blackman, MJ; Kvarnstrom, I; Jansson, K; Vrang, L; Hamelink, E; Hallberg, A; Rosenquist, A; Samuelsson, B Design and synthesis of potent inhibitors of the malaria aspartyl proteases plasmepsin I and II. Use of solid-phase synthesis to explore novel statine motifs. J Med Chem 47:3353-66 (2004) [PubMed] Article More Info.:
Target
Name:
Plasmepsin I
Synonyms:
PFAPG | PLM1_PLAFX | PMI | Plasmepsin 1 | Plasmepsin-1 precursor
Type:
Enzyme
Mol. Mass.:
51463.30
Organism:
Plasmodium falciparum
Description:
n/a
Residue:
452
Sequence:
MALSIKEDFSSAFAKNESAVNSSTFNNNMKTWKIQKRFQILYVFFFLLITGALFYYLIDNVLFPKNKKINEIMNTSKHVIIGFSIENSHDRIMKTVKQHRLKNYIKESLKFFKTGLTQKPHLGNAGDSVTLNDVANVMYYGEAQIGDNKQKFAFIFDTGSANLWVPSAQCNTIGCKTKNLYDSNKSKTYEKDGTKVEMNYVSGTVSGFFSKDIVTIANLSFPYKFIEVTDTNGFEPAYTLGQFDGIVGLGWKDLSIGSVDPVVVELKNQNKIEQAVFTFYLPFDDKHKGYLTIGGIEDRFYEGQLTYEKLNHDLYWQVDLDLHFGNLTVEKATAIVDSGTSSITAPTEFLNKFFEGLDVVKIPFLPLYITTCNNPKLPTLEFRSATNVYTLEPEYYLQQIFDFGISLCMVSIIPVDLNKNTFILGDPFMRKYFTVFDYDNHTVGFALAKKKL
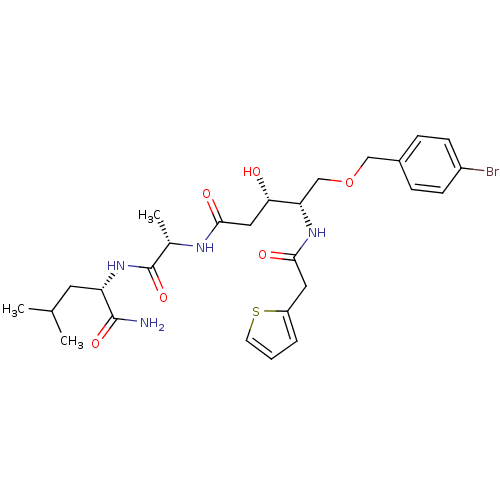
Inhibitor
Name:
BDBM7991
Synonyms:
(3S,4S)-5-(4-Bromobenzyloxy)-3-hydroxy-4-(2-thiophen-2-ylacetylamino)pentanoic Acid [(S)-1-((S)-1-Carbamoyl-3-methylbutylcarbamoyl)ethyl]amide | (3S,4S)-5-[(4-bromophenyl)methoxy]-N-[(1S)-1-{[(1S)-1-carbamoyl-3-methylbutyl]carbamoyl}ethyl]-3-hydroxy-4-[1-(thiophen-2-yl)acetamido]pentanamide | Statine-like inhibitor 30
Type:
Small organic molecule
Emp. Form.:
C27H37BrN4O6S
Mol. Mass.:
625.575
SMILES:
CC(C)C[C@H](NC(=O)[C@H](C)NC(=O)C[C@H](O)[C@H](COCc1ccc(Br)cc1)NC(=O)Cc1cccs1)C(N)=O |r|
Substrate
Name:
Peptide Substrate
Synonyms:
n/a
Type:
Peptide
Mol. Mass.:
3501.67
Organism:
n/a
Description:
n/a
Residue:
31
Sequence:
DACYLGLARGNLEPHELESERPHEPREDANS