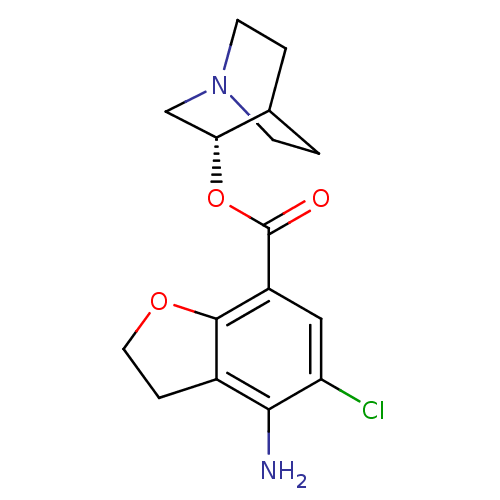
BDBM50288590 4-Amino-5-chloro-2,3-dihydro-benzofuran-7-carboxylic acid (S)-(1-aza-bicyclo[2.2.2]oct-3-yl) ester::CHEMBL92100
SMILES Nc1c2CCOc2c(cc1Cl)C(=O)O[C@@H]1CN2CCC1CC2
InChI Key InChIKey=QUSHSFRRPJRJKS-CYBMUJFWSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50288590
Found 3 hits for monomerid = 50288590
Affinity DataKi: 0.200nMAssay Description:Binding affinity at 5-hydroxytryptamine 3 receptor in rat entorhinal cortex by [3H]BRL-43694 displacement.More data for this Ligand-Target Pair
Affinity DataKi: 8.30nMAssay Description:Binding affinity at 5-hydroxytryptamine 4 receptor in rat striatum by [3H]GR-113808 displacement.More data for this Ligand-Target Pair
Affinity DataEC50: 11nMAssay Description:In vitro relaxation of carbachol pre-contracted rat oesophageal TMM.More data for this Ligand-Target Pair