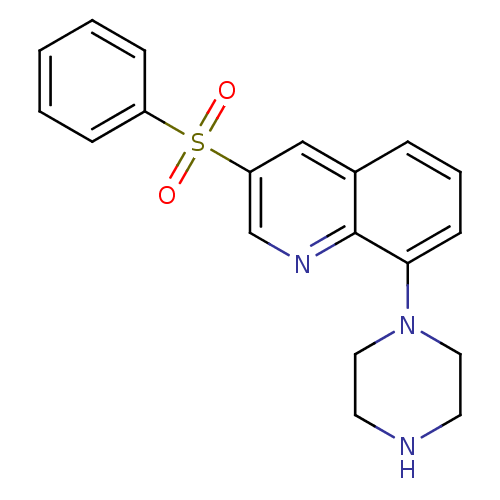
BDBM50318633 3-benzenesulfonyl-8-piperazin-1-ylquinoline::CHEMBL1083390::SB-742457
SMILES O=S(=O)(c1ccccc1)c1cnc2c(cccc2c1)N1CCNCC1
InChI Key InChIKey=JJZFWROHYSMCMU-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 25 hits for monomerid = 50318633
Found 25 hits for monomerid = 50318633
Affinity DataKi: 0.170nMAssay Description:Displacement of [3H]LSD from human 5-HT6 receptor expressed in human HeLa cells after 1 hr by scintillation spectroscopic analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.230nMAssay Description:Antagonist activity at 5-HT6 receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 0.230nMAssay Description:Inhibitory concentration against Herpes simplex virus type 1 thymidine kinase(HSV-1 TK)More data for this Ligand-Target Pair
Affinity DataKi: 0.234nMAssay Description:Binding affinity to 5HT6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.234nMAssay Description:Displacement of [3H]Lu AE60157 from rat brain 5-HT6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.234nMAssay Description:Displacement of [3H]Lu AE60157 from rat brain 5-HT6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.25nMAssay Description:Binding affinity to 5-HT6R (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataKi: 0.25nMAssay Description:Antagonist activity at human recombinant 5HT6 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.280nMAssay Description:Binding affinity to 5HT6R (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]-LSD from human 5HT6 receptor expressed in HEK293 cell membranes measured after 1 hr by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]LSD from human 5-HT6R expressed in HEK293 cell membranes after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]-LSD from human 5HT6R expressed in HEK293 cell membranes incubated for 1 hr by micro-beta plate reader based analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.40nMAssay Description:Displacement of [3H]-LSD from human 5HT6R expressed in HEK293 cell membranes after 1 hr by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Displacement of [3H]-8-OH-DPAT from human 5HT2A expressed in HEK293 cell membranes incubated for 1 hr by micro-beta plate reader based analysisMore data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Displacement of [3H]-Ketanserin from human 5HT2AR expressed in CHO-K1 cell membranes after 1.5 hrs by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 997nMAssay Description:Displacement of [3H]-raclopride from human D2L expressed in CHO-K1 cell membranes incubated for 1 hr by micro-beta plate reader based analysisMore data for this Ligand-Target Pair
Affinity DataKi: 997nMAssay Description:Displacement of [3H]-Raclopride from human dopamine D2L receptor expressed in HEK293 cell membranes after 1 hr by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 2.37E+3nMAssay Description:Displacement of [3H]-8-OH-DPAT from human 5HT1AR expressed in HEK293 cell membranes after 1 hr by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 2.37E+3nMAssay Description:Displacement of [3H]-8-OH-DPAT from human 5HT1A expressed in HEK293 cell membranes incubated for 1 hr by micro-beta plate reader based analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.42E+4nMAssay Description:Displacement of [3H]-5-CT from human 5HT7b expressed in HEK293 cell membranes incubated for 1 hr by micro-beta plate reader based analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.42E+4nMAssay Description:Displacement of [3H]-5-CT from human 5HT7BR expressed in HEK293 cell membranes after 1 hr by microbeta counting methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.80nMAssay Description:Antagonist activity at 5-HT6 receptor (unknown origin) expressed in NG108-15 cells assessed as reduction in cAMP level after 5 mins by BRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 97nMAssay Description:Inverse agonist activity at recombinant human 5HT6 receptor expressed in NG108-15 cells assessed as inhibition of cAMP production measured after 5 mi...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80nMAssay Description:Antagonist activity at 5HT6R (unknown origin) transfected in NG108-15 cells co-transfected with CAMYEL assessed as reduction in cAMP levels after 5 m...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80nMAssay Description:Inverse agonist activity at 5HT6R (unknown origin) expressed in 1321N1 cells co-expressing CAMYEL assessed as reduction in basal cAMP accumulation by...More data for this Ligand-Target Pair