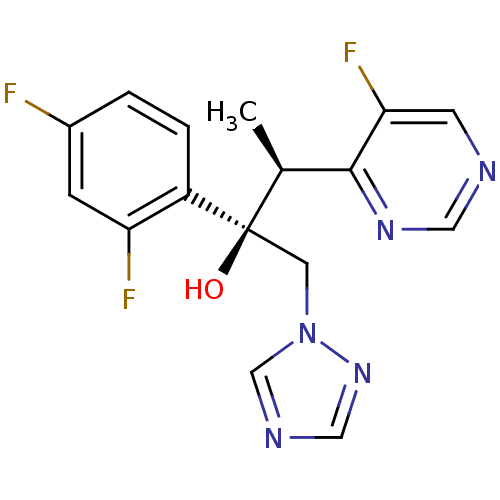
BDBM50333117 (2R,3S)-2,3-bis(2,4-difluorophenyl)-1-(1H-1,2,4-triazol-1-yl)butan-2-ol::(2R,3S)-2-(2,4-Difluoro-phenyl)-3-(5-fluoro-pyrimidin-4-yl)-1-[1,2,4]triazol-1-yl-butan-2-ol::(2R,3S)-2-(2,4-difluorophenyl)-3-(5-fluoropyrimidin-4-yl)-1-(1H-1,2,4-triazol-1-yl)butan-2-ol::CHEMBL638::UK-109496::US11247981, Example Voriconazole::US9221791, Voriconazole::VORICONAZOLE::Vfend
SMILES C[C@@H](c1ncncc1F)[C@](O)(Cn1cncn1)c1ccc(F)cc1F
InChI Key InChIKey=BCEHBSKCWLPMDN-MGPLVRAMSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50333117
Found 4 hits for monomerid = 50333117
Affinity DataKi: 340nMAssay Description:Inhibition of CYP2B6 in human liver microsomes assessed as bupropion 4-hydroxylation after 15 mins by Dixon plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Inhibition of CYP2B6 in human liver microsomes assessed as efavirenz 8-hydroxylation after 10 mins by Dixon plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 2.80E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomes assessed as tolbutamide 4-methylhydroxylation after 60 mins by Dixon plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 5.10E+3nMAssay Description:Inhibition of CYP2C19 in human liver microsomes assessed as S-mephenytoin 4'-hydroxylation after 60 mins by Dixon plot analysisMore data for this Ligand-Target Pair