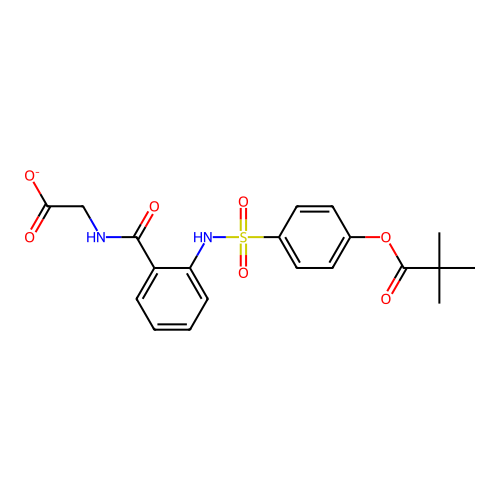
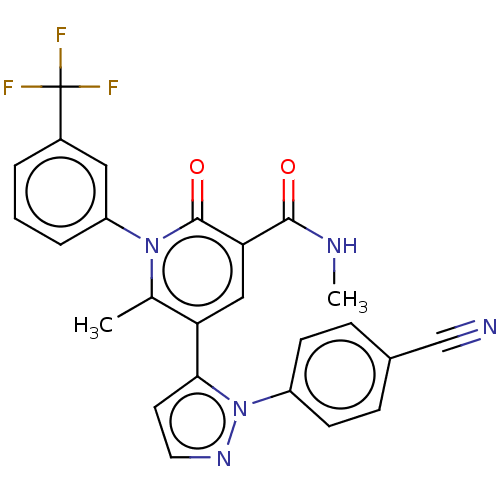
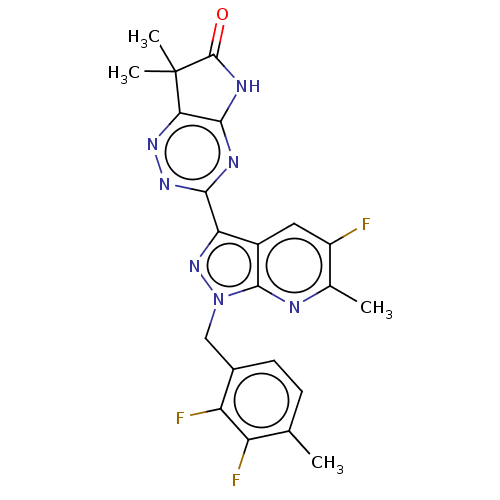
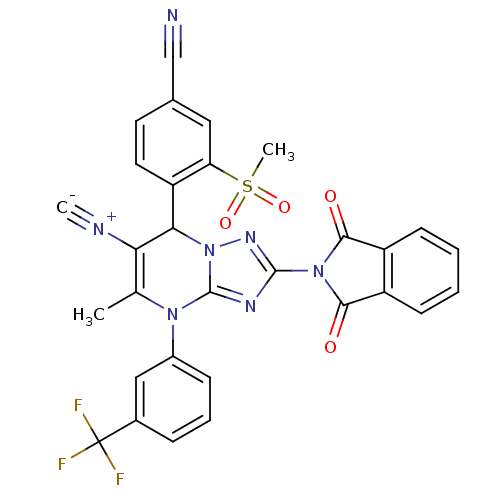
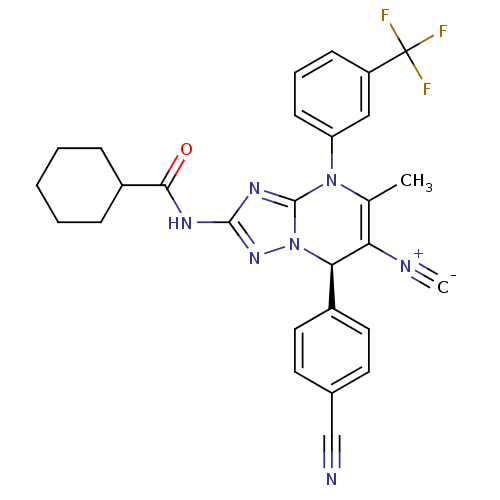
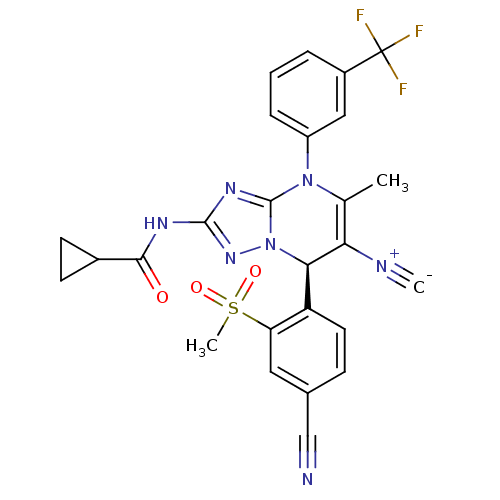
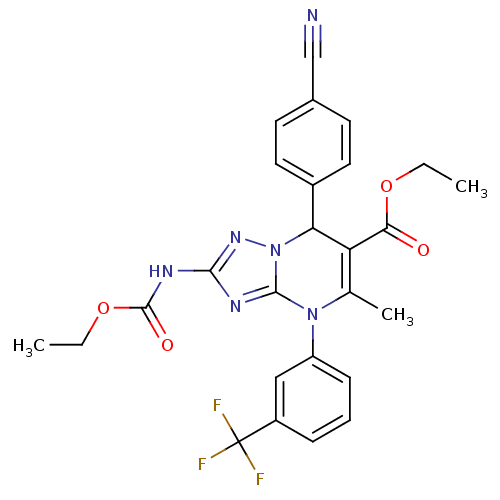
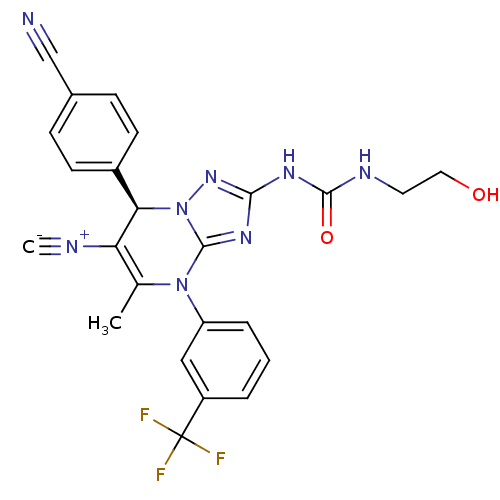
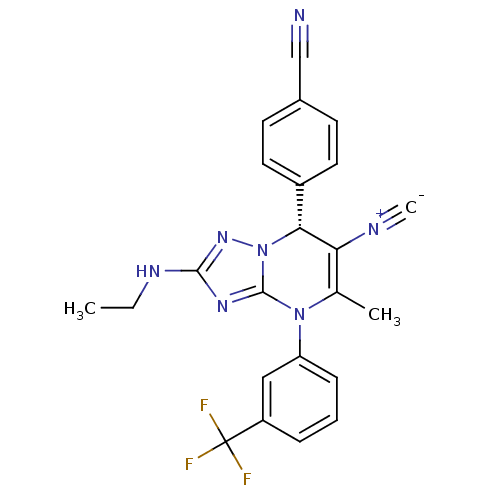
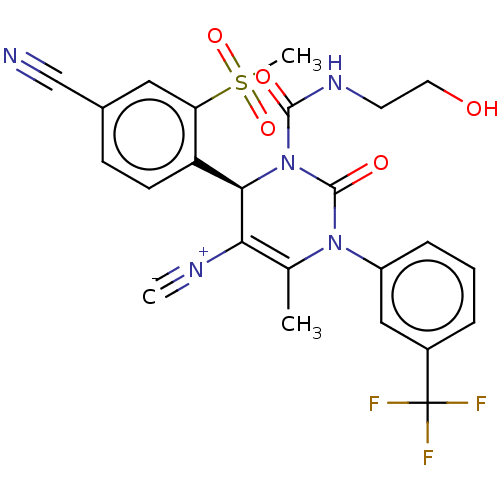
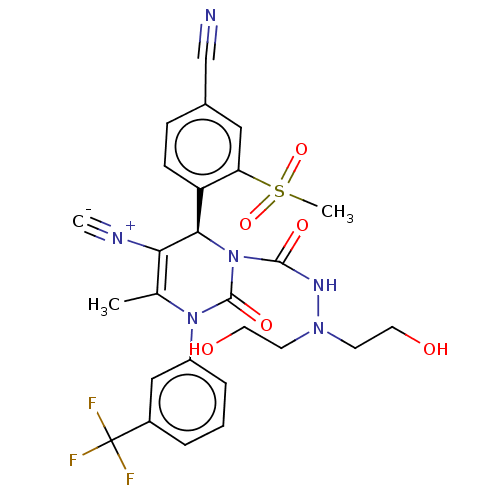
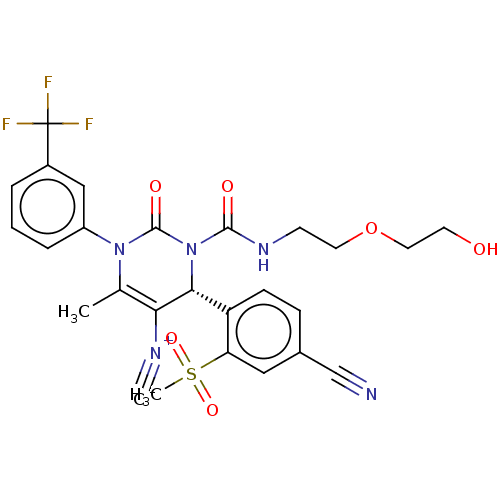
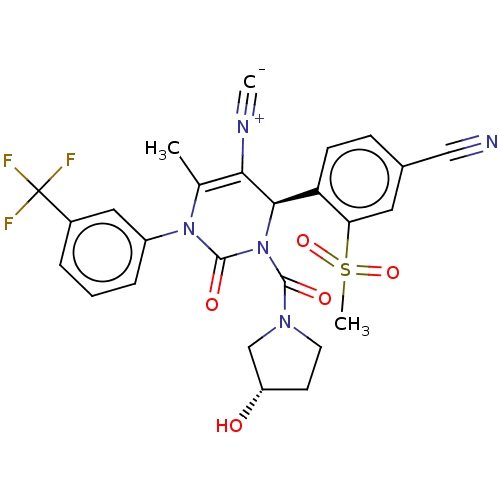
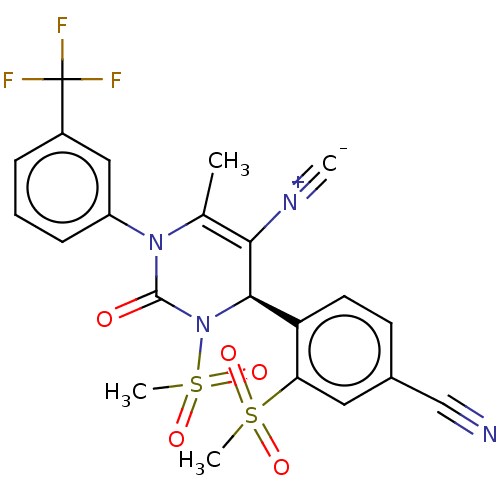
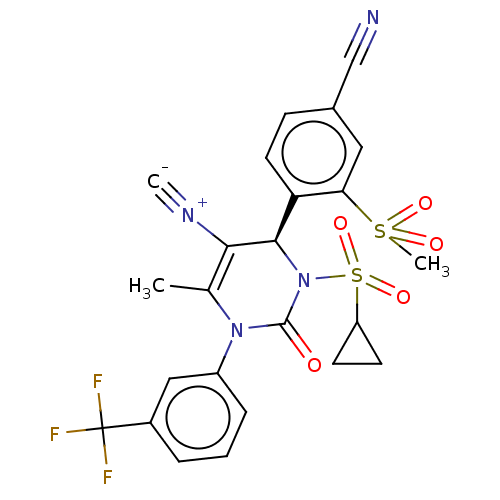
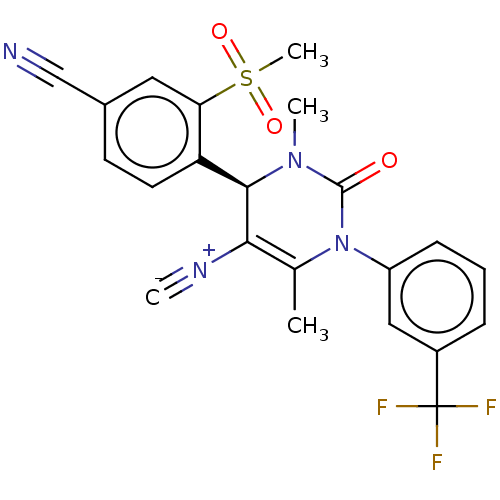
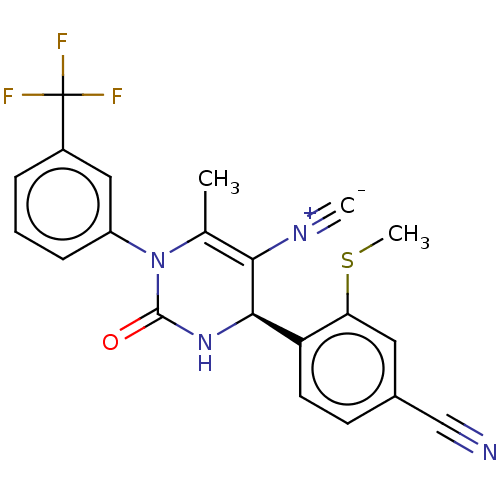
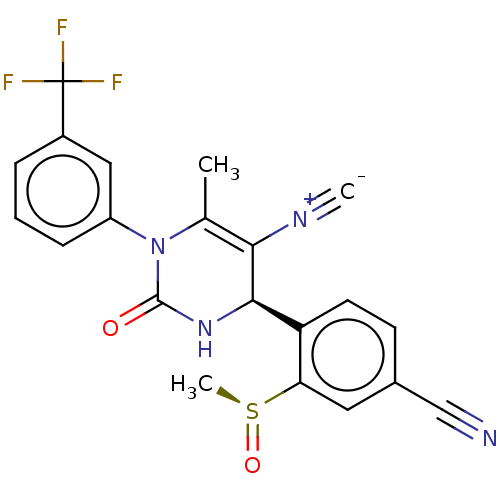
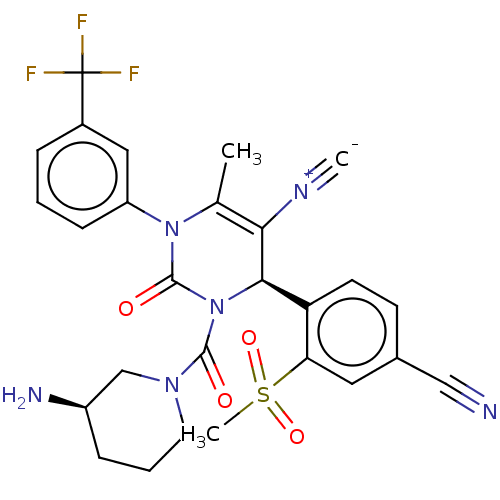
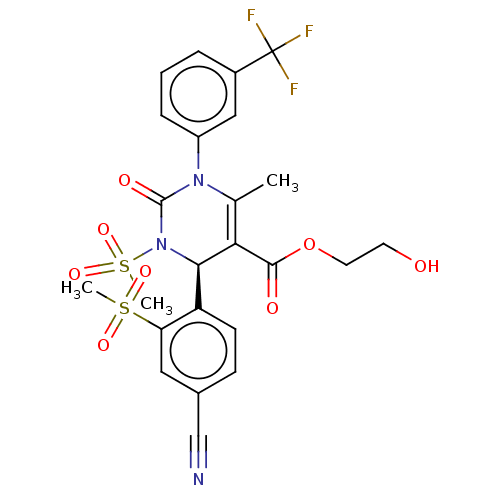
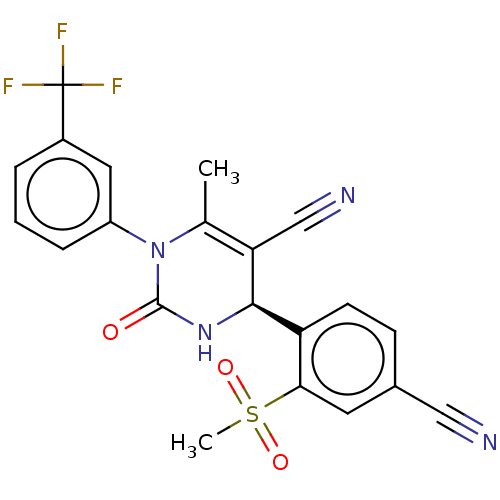
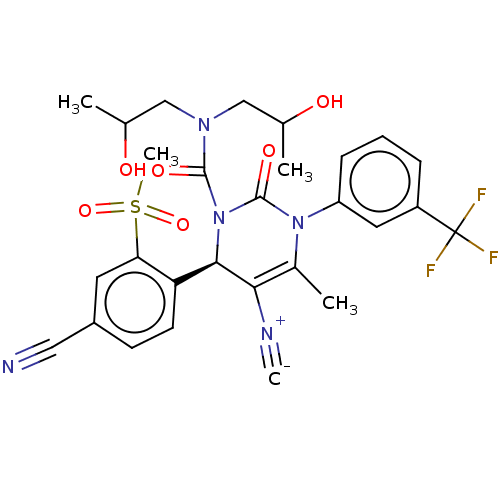
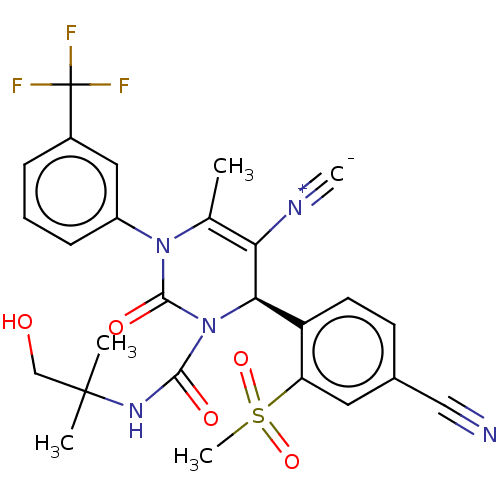
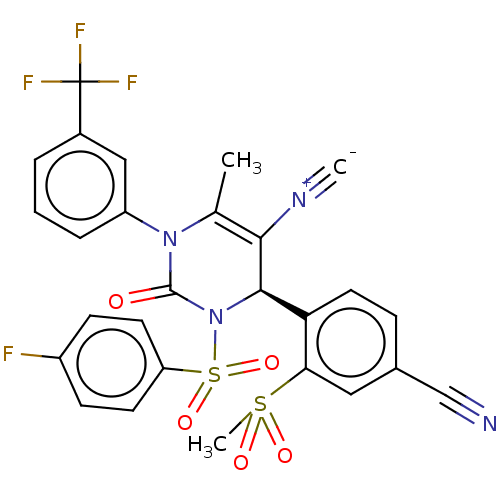
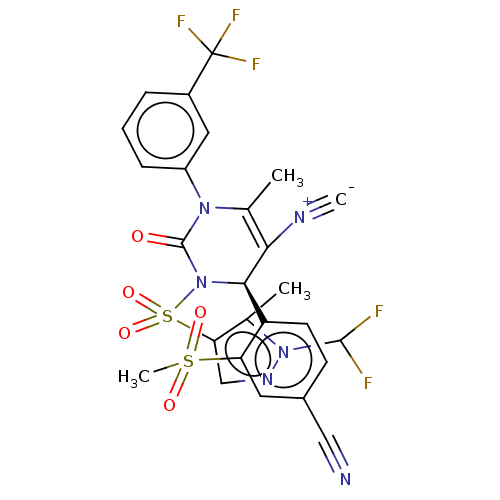
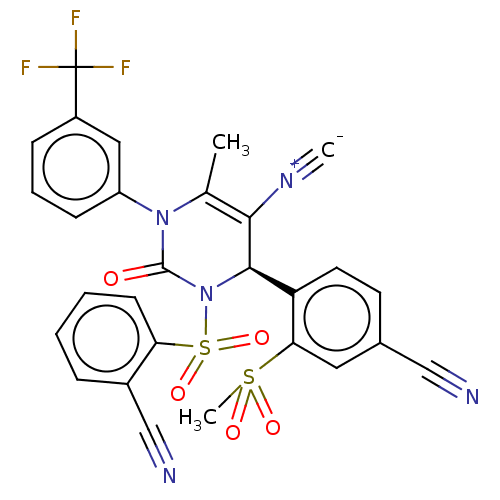
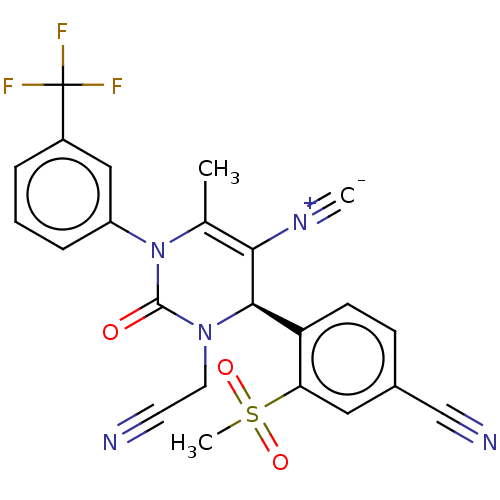
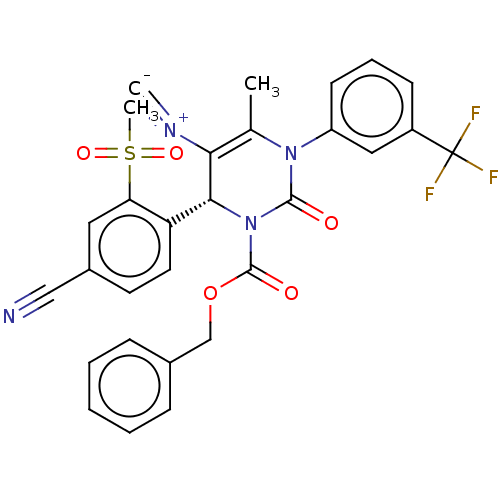
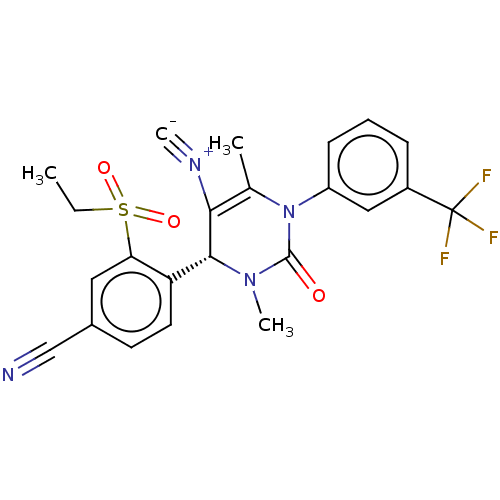
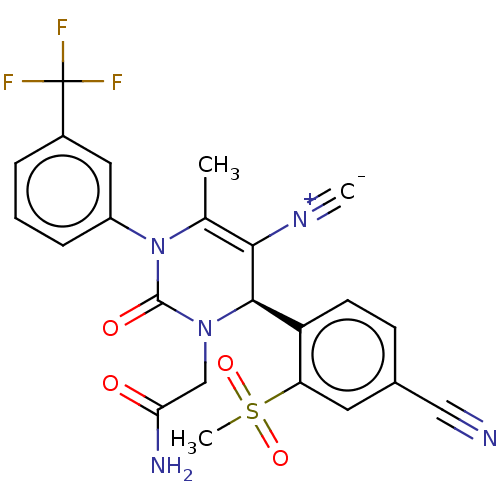
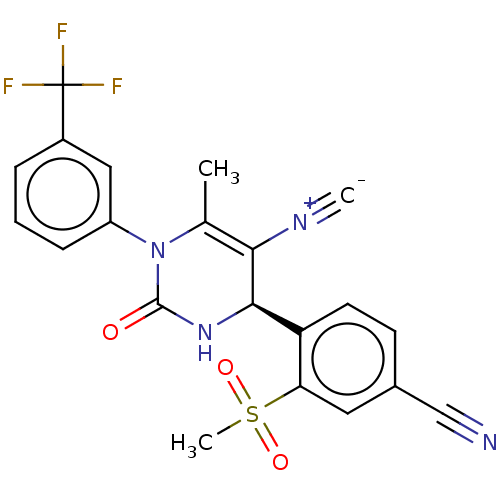
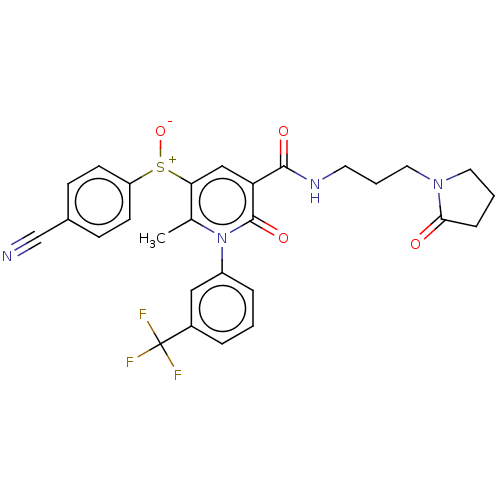
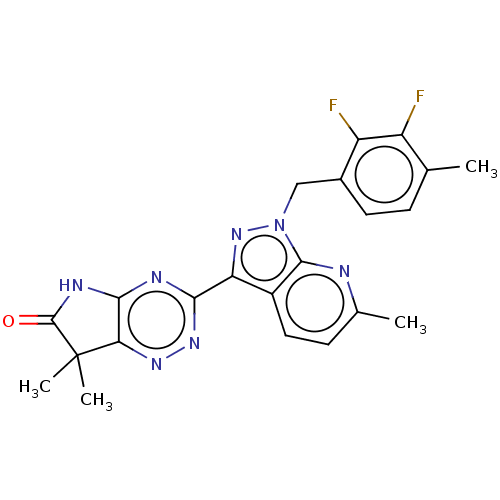
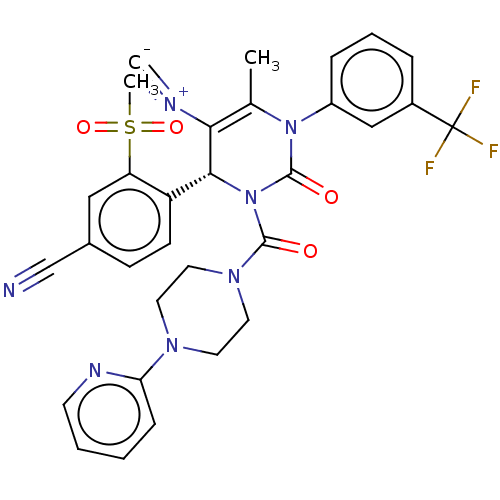
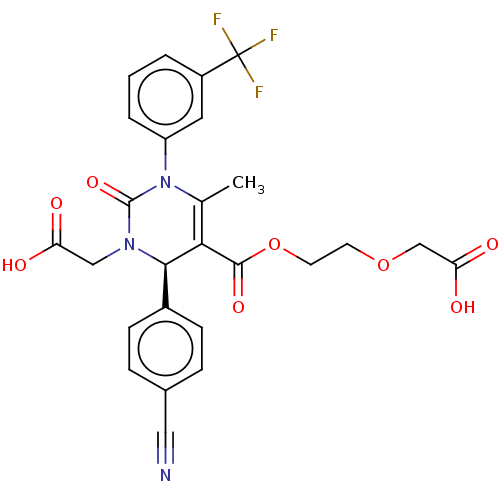
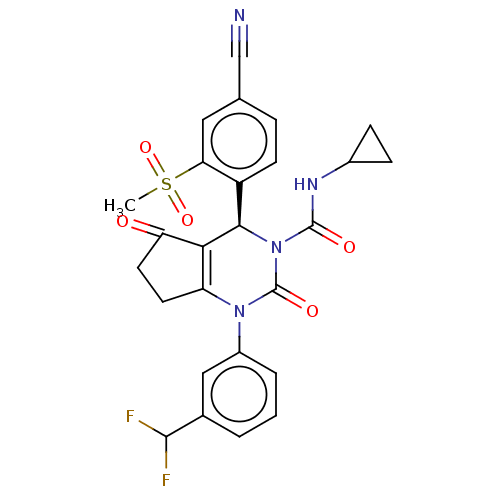
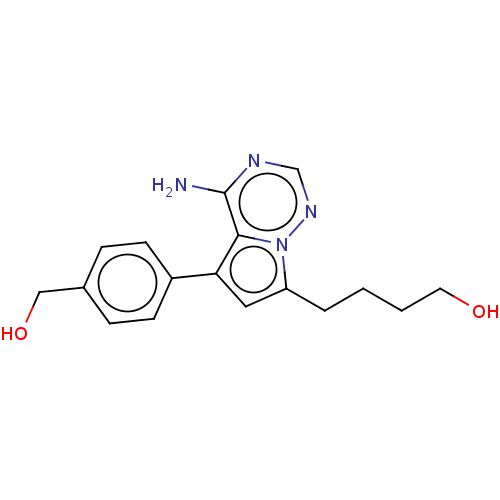
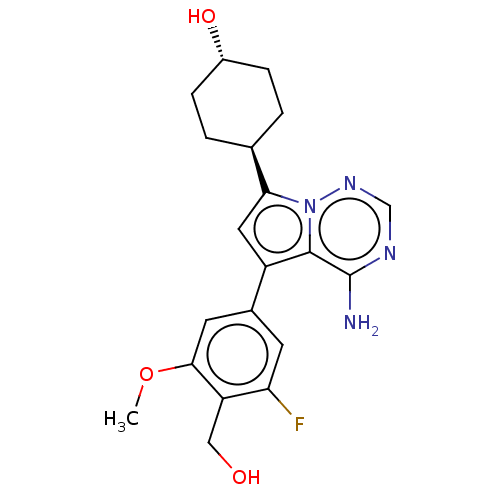
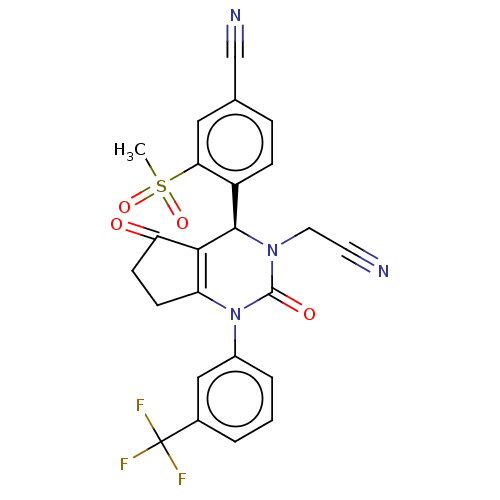
Affinity DataKi: 0.0800nMAssay Description:Inhibition of human neutrophil elastase using MeOSuc-AAPV-AMC as substrate after 60 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.160nMAssay Description:Inhibition of neutrophil elastase in human whole blood using MeO-Succ-Ala-Ala-Pro-Val-pNA as substrate after 30 mins by colorimetric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Inhibition of neutrophil elastase in human whole blood using MeO-Succ-Ala-Ala-Pro-Val-pNA as substrate after 30 mins by colorimetric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Inhibition of human neutrophil elastase using MeO-Suc-Ala-Ala-Pro-Val 7-amido-4-methylcoumarin as substrate preincubated for 15 mins followed by subs...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Inhibition of human neutrophil elastase using MeOSuc-AAPV-AMC as substrate after 60 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 200nMAssay Description:Inhibition of human neutrophil elastase using suc-Ala-Pro-Ala-pNA as substrate after 30 mins by spectrophotometric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.40E+3nMAssay Description:Inhibition of neutrophil elastase in human whole blood using MeO-Succ-Ala-Ala-Pro-Val-pNA as substrate after 30 mins by colorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Bayer Pharma Aktiengesellschaft
US Patent
Bayer Pharma Aktiengesellschaft
US Patent
Affinity DataIC50: 0.25nMAssay Description:To determine their in vitro action on human PDE 5, the test substances are dissolved in 100% DMSO and serially diluted. Typically, dilution series (1...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:In vitro HNE inhibition assay. The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amido...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMAssay Description:Inhibition of human neutrophil elastase using MeOSuc-AAPV-AMC as substrate after 60 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: <0.300nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: 0.450nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
TargetcGMP-specific 3',5'-cyclic phosphodiesterase(Homo sapiens (Human))
Bayer Pharma Aktiengesellschaft
US Patent
Bayer Pharma Aktiengesellschaft
US Patent
Affinity DataIC50: 0.650nMAssay Description:To determine their in vitro action on human PDE 5, the test substances are dissolved in 100% DMSO and serially diluted. Typically, dilution series (1...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMpH: 7.4 T: 2°CAssay Description:The potency of the compounds of the invention is ascertained in an in vitro inhibition assay. The HNE-mediated amidolytic cleavage of a suitable pept...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:Inhibition of human neutrophil elastaseMore data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of neutrophil elastase in human plasma using MeOSuc-Ala-Ala-Pro-Val-AMC as substrate by fluorescence assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase receptor R3(Homo sapiens (Human))
Bayer Intellectual Property
US Patent
Bayer Intellectual Property
US Patent
Affinity DataIC50: 1nMAssay Description:In vitro enzyme inhibition using scintillation of incorporated radio label (flashplate assay). Test compounds diluted in DMSO are mixed with a suitab...More data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase receptor R3(Homo sapiens (Human))
Bayer Intellectual Property
US Patent
Bayer Intellectual Property
US Patent
Affinity DataIC50: 1nMAssay Description:In vitro enzyme inhibition using scintillation of incorporated radio label (flashplate assay). Test compounds diluted in DMSO are mixed with a suitab...More data for this Ligand-Target Pair
Affinity DataIC50: <1nMAssay Description:Inhibition of human neutrophil elastase using MeO-Succ-Ala-Ala-Pro-Val-AMC as substrate after 30 mins by fluorescence assayMore data for this Ligand-Target Pair


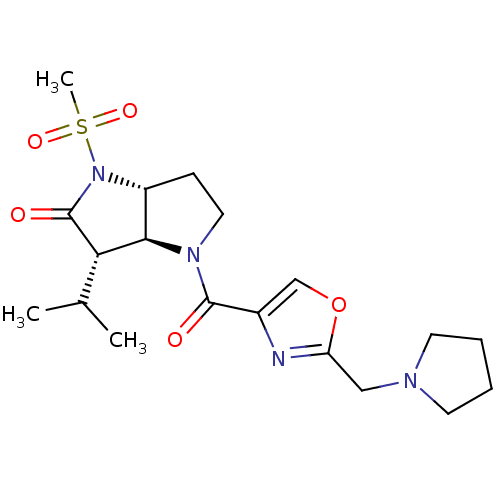
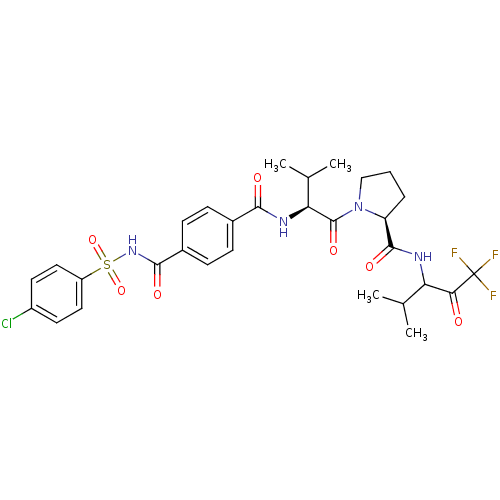
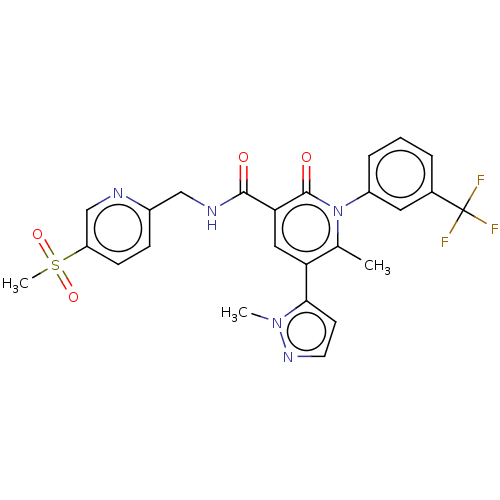
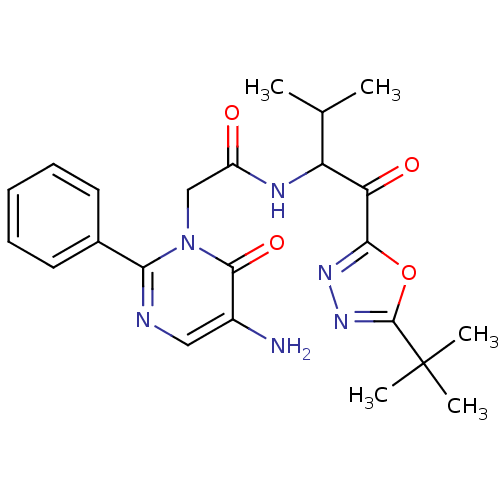
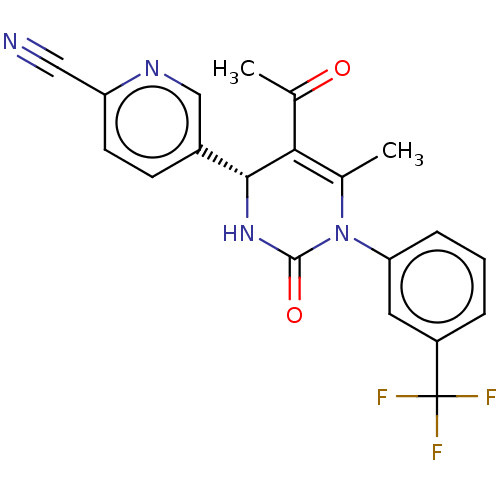
 3D Structure (crystal)
3D Structure (crystal)