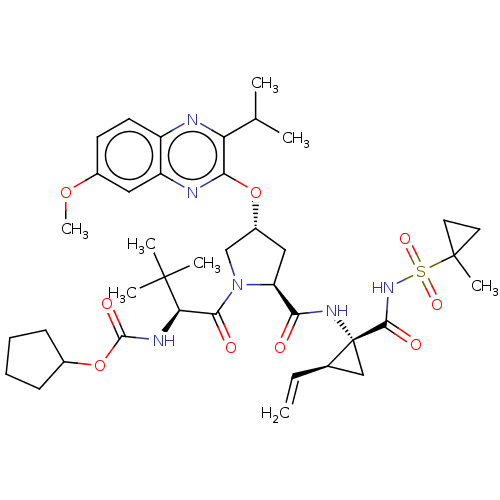
TargetGenome polyprotein/Non-structural protein 4A(Hepatitis C virus)
University Of Massachusetts Medical School
Curated by ChEMBL
University Of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: 21nMAssay Description:Inhibition of wild type HCV genotype 1a NS3/4A protease expressed in Escherichia coli BL21(DE3) using Ac-DE-Dap(QXL 520)-EE-Abu-psi-[COO]AS-C(5-FAMsp...More data for this Ligand-Target Pair
TargetGenome polyprotein/Non-structural protein 4A(Hepatitis C virus)
University Of Massachusetts Medical School
Curated by ChEMBL
University Of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: 43nMAssay Description:Inhibition of HCV genotype 1a NS3/4A protease R155K mutant expressed in Escherichia coli BL21(DE3) using Ac-DE-Dap(QXL 520)-EE-Abu-psi-[COO]AS-C(5-FA...More data for this Ligand-Target Pair
TargetGenome polyprotein/Non-structural protein 4A(Hepatitis C virus)
University Of Massachusetts Medical School
Curated by ChEMBL
University Of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Inhibition of HCV genotype 1a NS3/4A protease D168A mutant expressed in Escherichia coli BL21(DE3) using Ac-DE-Dap(QXL 520)-EE-Abu-psi-[COO]AS-C(5-FA...More data for this Ligand-Target Pair

 3D Structure (crystal)
3D Structure (crystal)