Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
3-hydroxy-3-methylglutaryl-coenzyme A reductase
Ligand
BDBM18372
Substrate
BDBM18373
Meas. Tech.
HMG-CoA Reductase In Vitro Assay
pH
7.2±n/a
Temperature
310.15±n/a K
IC50
3.6±n/a nM
Citation
 Bratton, LD; Auerbach, B; Choi, C; Dillon, L; Hanselman, JC; Larsen, SD; Lu, G; Olsen, K; Pfefferkorn, JA; Robertson, A; Sekerke, C; Trivedi, BK; Unangst, PC Discovery of pyrrole-based hepatoselective ligands as potent inhibitors of HMG-CoA reductase. Bioorg Med Chem 15:5576-89 (2007) [PubMed] Article
Bratton, LD; Auerbach, B; Choi, C; Dillon, L; Hanselman, JC; Larsen, SD; Lu, G; Olsen, K; Pfefferkorn, JA; Robertson, A; Sekerke, C; Trivedi, BK; Unangst, PC Discovery of pyrrole-based hepatoselective ligands as potent inhibitors of HMG-CoA reductase. Bioorg Med Chem 15:5576-89 (2007) [PubMed] Article More Info.:
Target
Name:
3-hydroxy-3-methylglutaryl-coenzyme A reductase
Synonyms:
HMDH_RAT | HMG-CoA reductase | Hmgcr
Type:
Enzyme
Mol. Mass.:
96689.85
Organism:
Rattus norvegicus (rat)
Description:
Isolated rat liver microsomes were used as enzyme source.
Residue:
887
Sequence:
MLSRLFRMHGLFVASHPWEVIVGTVTLTICMMSMNMFTGNNKICGWNYECPKFEEDVLSSDIIILTITRCIAILYIYFQFQNLRQLGSKYILGIAGLFTIFSSFVFSTVVIHFLDKELTGLNEALPFFLLLIDLSRASALAKFALSSNSQDEVRENIARGMAILGPTFTLDALVECLVIGVGTMSGVRQLEIMCCFGCMSVLANYFVFMTFFPACVSLVLELSRESREGRPIWQLSHFARVLEEEENKPNPVTQRVKMIMSLGLVLVHAHSRWIADPSPQNSTAEQSKVSLGLAEDVSKRIEPSVSLWQFYLSKMISMDIEQVITLSLALLLAVKYIFFEQAETESTLSLKNPITSPVVTPKKAQDNCCRREPLLVRRNQKLSSVEEDPGVNQDRKVEVIKPLVAEAETSGRATFVLGASAASPPLALGAQEPGIELPSEPRPNEECLQILESAEKGAKFLSDAEIIQLVNAKHIPAYKLETLMETHERGVSIRRQLLSAKLAEPSSLQYLPYRDYNYSLVMGACCENVIGYMPIPVGVAGPLCLDGKEYQVPMATTEGCLVASTNRGCRAISLGGGASSRVLADGMSRGPVVRLPRACDSAEVKSWLETPEGFAVVKEAFDSTSRFARLQKLHVTLAGRNLYIRLQSKTGDAMGMNMISKGTEKALLKLQEFFPELQILAVSGNYCTDKKPAAINWIEGRGKTVVCEAVIPAKVVREVLKTTTEAMVDVNINKNLVGSAMAGSIGGYNAHAANIVTAIYIACGQDAAQNVGSSNCITLMEASGPTNEDLYISCTMPSIEIGTVGGGTNLLPQQACLQMLGVQGACKDNPGENARQLARIVCGTVMAGELSLMAALAAGHLVRSHMVHNRSKINLQDLQGTCTKKAA
Inhibitor
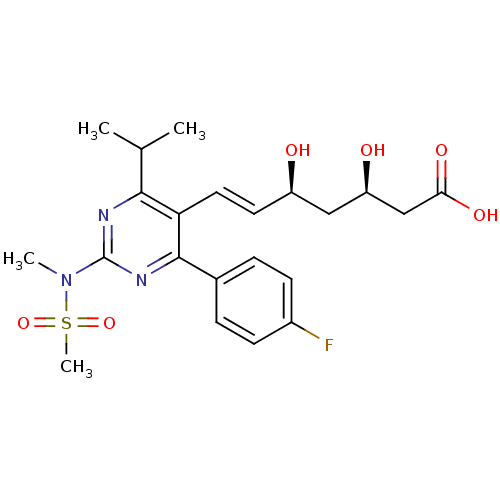
Name:
BDBM18372
Synonyms:
(3R,5S,6E)-7-[4-(4-fluorophenyl)-2-(N-methylmethanesulfonamido)-6-(propan-2-yl)pyrimidin-5-yl]-3,5-dihydroxyhept-6-enoic acid | CHEMBL1496 | Ros | Rosuvastatin | US9102656, Rosuvastatin | ZD4522
Type:
Small organic molecule
Emp. Form.:
C22H28FN3O6S
Mol. Mass.:
481.538
SMILES:
CC(C)c1nc(nc(-c2ccc(F)cc2)c1\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)N(C)S(C)(=O)=O |r|