Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Peroxisome proliferator-activated receptor alpha
Ligand
BDBM24566
Substrate
BDBM10852
Meas. Tech.
Cell-Based Transcription Assay
EC50
40±2 nM
Comments
100% efficacy.
Citation
 Montanari, R; Saccoccia, F; Scotti, E; Crestani, M; Godio, C; Gilardi, F; Loiodice, F; Fracchiolla, G; Laghezza, A; Tortorella, P; Lavecchia, A; Novellino, E; Mazza, F; Aschi, M; Pochetti, G Crystal structure of the peroxisome proliferator-activated receptor gamma (PPARgamma) ligand binding domain complexed with a novel partial agonist: a new region of the hydrophobic pocket could be exploited for drug design. J Med Chem 51:7768-76 (2008) [PubMed] Article
Montanari, R; Saccoccia, F; Scotti, E; Crestani, M; Godio, C; Gilardi, F; Loiodice, F; Fracchiolla, G; Laghezza, A; Tortorella, P; Lavecchia, A; Novellino, E; Mazza, F; Aschi, M; Pochetti, G Crystal structure of the peroxisome proliferator-activated receptor gamma (PPARgamma) ligand binding domain complexed with a novel partial agonist: a new region of the hydrophobic pocket could be exploited for drug design. J Med Chem 51:7768-76 (2008) [PubMed] Article More Info.:
Target
Name:
Peroxisome proliferator-activated receptor alpha
Synonyms:
Nr1c1 | Nuclear receptor subfamily 1 group C member 1 | PPAR-alpha | PPARA_MOUSE | Peroxisome Proliferator-Activated Receptor alpha | Ppar | Ppara
Type:
PROTEIN
Mol. Mass.:
52343.28
Organism:
Mus musculus
Description:
ChEMBL_572395
Residue:
468
Sequence:
MVDTESPICPLSPLEADDLESPLSEEFLQEMGNIQEISQSIGEESSGSFGFADYQYLGSCPGSEGSVITDTLSPASSPSSVSCPVIPASTDESPGSALNIECRICGDKASGYHYGVHACEGCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSEKAKLKAEILTCEHDLKDSETADLKSLGKRIHEAYLKNFNMNKVKARVILAGKTSNNPPFVIHDMETLCMAEKTLVAKMVANGVEDKEAEVRFFHCCQCMSVETVTELTEFAKAIPGFANLDLNDQVTLLKYGVYEAIFTMLSSLMNKDGMLIAYGNGFITREFLKNLRKPFCDIMEPKFDFAMKFNALELDDSDISLFVAAIICCGDRPGLLNIGYIEKLQEGIVHVLKLHLQSNHPDDTFLFPKLLQKMVDLRQLVTEHAQLVQVIKKTESDAALHPLLQEIYRDMY
Inhibitor
Name:
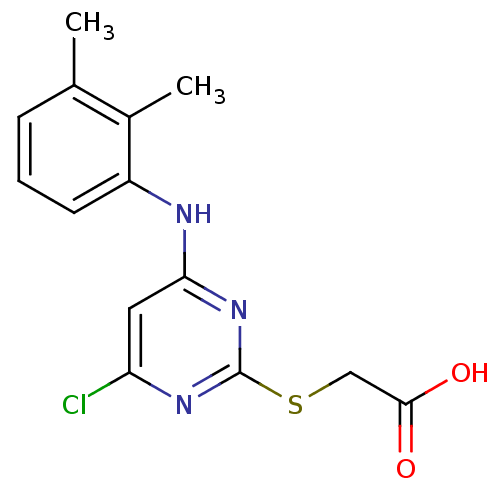
BDBM24566
Synonyms:
2-({4-chloro-6-[(2,3-dimethylphenyl)amino]pyrimidin-2-yl}sulfanyl)acetic acid | CHEMBL295416 | JMC515449 Compound 1 | Pirinixic acid | Wyeth 14,643
Type:
Small organic molecule
Emp. Form.:
C14H14ClN3O2S
Mol. Mass.:
323.798
SMILES:
Cc1cccc(Nc2cc(Cl)nc(SCC(O)=O)n2)c1C
Substrate