Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutamate receptor ionotropic, NMDA 2B
Ligand
BDBM50220725
Substrate
n/a
Meas. Tech.
ChEMBL_449356 (CHEMBL899623)
IC50
96±n/a nM
Citation
 Kawai, M; Nakamura, H; Sakurada, I; Shimokawa, H; Tanaka, H; Matsumizu, M; Ando, K; Hattori, K; Ohta, A; Nukui, S; Omura, A; Kawamura, M Discovery of novel and orally active NR2B-selective N-methyl-D-aspartate (NMDA) antagonists, pyridinol derivatives with reduced HERG binding affinity. Bioorg Med Chem Lett 17:5533-6 (2007) [PubMed] Article
Kawai, M; Nakamura, H; Sakurada, I; Shimokawa, H; Tanaka, H; Matsumizu, M; Ando, K; Hattori, K; Ohta, A; Nukui, S; Omura, A; Kawamura, M Discovery of novel and orally active NR2B-selective N-methyl-D-aspartate (NMDA) antagonists, pyridinol derivatives with reduced HERG binding affinity. Bioorg Med Chem Lett 17:5533-6 (2007) [PubMed] Article More Info.:
Target
Name:
Glutamate receptor ionotropic, NMDA 2B
Synonyms:
GluN2B | Glutamate [NMDA] receptor subunit epsilon 2 | Grin2b | N-methyl D-aspartate receptor subtype 2B | NMDA receptor subunit N2B (GluN2B) | NMDAR2B | NMDE2_RAT | NR2B
Type:
Protein
Mol. Mass.:
166077.66
Organism:
Rattus norvegicus (Rat)
Description:
Q00960
Residue:
1482
Sequence:
MKPSAECCSPKFWLVLAVLAVSGSKARSQKSPPSIGIAVILVGTSDEVAIKDAHEKDDFHHLSVVPRVELVAMNETDPKSIITRICDLMSDRKIQGVVFADDTDQEAIAQILDFISAQTLTPILGIHGGSSMIMADKDESSMFFQFGPSIEQQASVMLNIMEEYDWYIFSIVTTYFPGYQDFVNKIRSTIENSFVGWELEEVLLLDMSLDDGDSKIQNQLKKLQSPIILLYCTKEEATYIFEVANSVGLTGYGYTWIVPSLVAGDTDTVPSEFPTGLISVSYDEWDYGLPARVRDGIAIITTAASDMLSEHSFIPEPKSSCYNTHEKRIYQSNMLNRYLINVTFEGRNLSFSEDGYQMHPKLVIILLNKERKWERVGKWKDKSLQMKYYVWPRMCPETEEQEDDHLSIVTLEEAPFVIVESVDPLSGTCMRNTVPCQKRIISENKTDEEPGYIKKCCKGFCIDILKKISKSVKFTYDLYLVTNGKHGKKINGTWNGMIGEVVMKRAYMAVGSLTINEERSEVVDFSVPFIETGISVMVSRSNGTVSPSAFLEPFSADVWVMMFVMLLIVSAVAVFVFEYFSPVGYNRCLADGREPGGPSFTIGKAIWLLWGLVFNNSVPVQNPKGTTSKIMVSVWAFFAVIFLASYTANLAAFMIQEEYVDQVSGLSDKKFQRPNDFSPPFRFGTVPNGSTERNIRNNYAEMHAYMGKFNQRGVDDALLSLKTGKLDAFIYDAAVLNYMAGRDEGCKLVTIGSGKVFASTGYGIAIQKDSGWKRQVDLAILQLFGDGEMEELEALWLTGICHNEKNEVMSSQLDIDNMAGVFYMLGAAMALSLITFICEHLFYWQFRHCFMGVCSGKPGMVFSISRGIYSCIHGVAIEERQSVMNSPTATMNNTHSNILRLLRTAKNMANLSGVNGSPQSALDFIRRESSVYDISEHRRSFTHSDCKSYNNPPCEENLFSDYISEVERTFGNLQLKDSNVYQDHYHHHHRPHSIGSTSSIDGLYDCDNPPFTTQPRSISKKPLDIGLPSSKHSQLSDLYGKFSFKSDRYSGHDDLIRSDVSDISTHTVTYGNIEGNAAKRRKQQYKDSLKKRPASAKSRREFDEIELAYRRRPPRSPDHKRYFRDKEGLRDFYLDQFRTKENSPHWEHVDLTDIYKERSDDFKRDSVSGGGPCTNRSHLKHGTGEKHGVVGGVPAPWEKNLTNVDWEDRSGGNFCRSCPSKLHNYSSTVAGQNSGRQACIRCEACKKAGNLYDISEDNSLQELDQPAAPVAVTSNASSTKYPQSPTNSKAQKKNRNKLRRQHSYDTFVDLQKEEAALAPRSVSLKDKGRFMDGSPYAHMFEMPAGESSFANKSSVPTAGHHHNNPGSGYMLSKSLYPDRVTQNPFIPTFGDDQCLLHGSKSYFFRQPTVAGASKTRPDFRALVTNKPVVVTLHGAVPGRFQKDICIGNQSNPCVPNNKNPRAFNGSSNGHVYEKLSSIESDV
Inhibitor
Name:
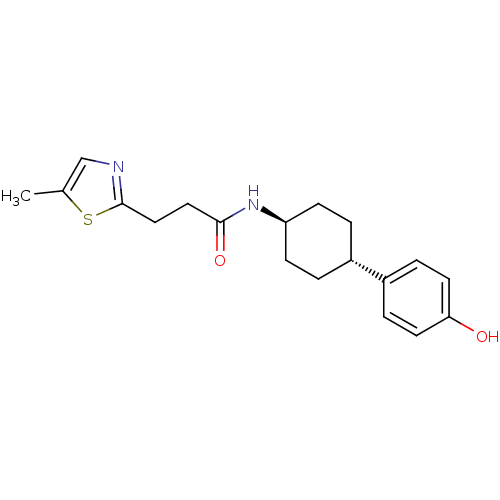
BDBM50220725
Synonyms:
CHEMBL237101 | N-((1r,4r)-4-(4-hydroxyphenyl)cyclohexyl)-3-(5-methylthiazol-2-yl)propanamide
Type:
Small organic molecule
Emp. Form.:
C19H24N2O2S
Mol. Mass.:
344.471
SMILES:
Cc1cnc(CCC(=O)N[C@H]2CC[C@@H](CC2)c2ccc(O)cc2)s1 |wU:13.16,wD:10.9,(9.08,-7.45,;7.55,-7.45,;6.65,-6.21,;5.19,-6.67,;5.18,-8.21,;3.84,-8.97,;2.51,-8.19,;1.18,-8.95,;1.18,-10.5,;-.16,-8.18,;-1.49,-8.96,;-2.83,-8.17,;-4.16,-8.96,;-4.16,-10.5,;-2.83,-11.27,;-1.49,-10.5,;-5.49,-11.27,;-6.83,-10.5,;-8.15,-11.26,;-8.16,-12.82,;-9.48,-13.59,;-6.81,-13.58,;-5.48,-12.81,;6.64,-8.7,)|