Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Adenosine deaminase
Ligand
BDBM14487
Substrate
n/a
Meas. Tech.
ChEMBL_31099 (CHEMBL640455)
Ki
2180000±n/a nM
Citation
 Bussolari, JC; Ramesh, K; Stoeckler, JD; Chen, SF; Panzica, RP Synthesis and biological evaluation of N4-substituted imidazo- and v-triazolo[4,5-d]pyridazine nucleosides. J Med Chem 36:4113-20 (1994) [PubMed] Article
Bussolari, JC; Ramesh, K; Stoeckler, JD; Chen, SF; Panzica, RP Synthesis and biological evaluation of N4-substituted imidazo- and v-triazolo[4,5-d]pyridazine nucleosides. J Med Chem 36:4113-20 (1994) [PubMed] Article More Info.:
Target
Name:
Adenosine deaminase
Synonyms:
ADA | ADA1 | ADA_HUMAN | Adenosine aminohydrolase
Type:
Enzyme
Mol. Mass.:
40759.44
Organism:
Homo sapiens (Human)
Description:
Human recombinant ADA was expressed and purified from an ADA-deficient bacterial strain.
Residue:
363
Sequence:
MAQTPAFDKPKVELHVHLDGSIKPETILYYGRRRGIALPANTAEGLLNVIGMDKPLTLPDFLAKFDYYMPAIAGCREAIKRIAYEFVEMKAKEGVVYVEVRYSPHLLANSKVEPIPWNQAEGDLTPDEVVALVGQGLQEGERDFGVKARSILCCMRHQPNWSPKVVELCKKYQQQTVVAIDLAGDETIPGSSLLPGHVQAYQEAVKSGIHRTVHAGEVGSAEVVKEAVDILKTERLGHGYHTLEDQALYNRLRQENMHFEICPWSSYLTGAWKPDTEHAVIRLKNDQANYSLNTDDPLIFKSTLDTDYQMTKRDMGFTEEEFKRLNINAAKSSFLPEDEKRELLDLLYKAYGMPPSASAGQNL
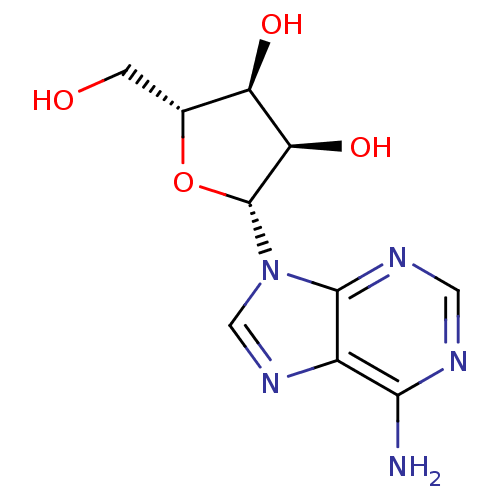
Inhibitor
Name:
BDBM14487
Synonyms:
(2R,3R,4S,5R)-2-(6-amino-9H-purin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol | Adenine-beta-D-arabinofuranoside | Adenosine | CHEMBL477 | N6-Methylado | [U-14C]adenosine | cid_191 | cid_60961
Type:
Nucleoside or nucleotide
Emp. Form.:
C10H13N5O4
Mol. Mass.:
267.2413
SMILES:
Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O