Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 5A
Ligand
BDBM135794
Substrate
n/a
Meas. Tech.
Binding Inhibition Assay
pH
7.5±n/a
Temperature
298.15±n/a K
Ki
4.6±n/a nM
Comments
extracted
Citation
 Kinoyama, I; Miyazaki, T; Koganemaru, Y; Washio, T; Hamaguchi, W Nitrogenous-ring acylguanidine derivative US Patent US8853242 Publication Date 10/7/2014
Kinoyama, I; Miyazaki, T; Koganemaru, Y; Washio, T; Hamaguchi, W Nitrogenous-ring acylguanidine derivative US Patent US8853242 Publication Date 10/7/2014 More Info.:
Target
Name:
5-hydroxytryptamine receptor 5A
Synonyms:
5-HT-5 | 5-HT-5A | 5-hydroxytryptamine receptor 5 (5-HT5) | 5-hydroxytryptamine receptor 5A (5-HT5A) | 5HT5A_HUMAN | HTR5A | Serotonin (5-HT) receptor | Serotonin receptor 5A
Type:
Enzyme
Mol. Mass.:
40266.25
Organism:
Homo sapiens (Human)
Description:
P47898
Residue:
357
Sequence:
MDLPVNLTSFSLSTPSPLETNHSLGKDDLRPSSPLLSVFGVLILTLLGFLVAATFAWNLLVLATILRVRTFHRVPHNLVASMAVSDVLVAALVMPLSLVHELSGRRWQLGRRLCQLWIACDVLCCTASIWNVTAIALDRYWSITRHMEYTLRTRKCVSNVMIALTWALSAVISLAPLLFGWGETYSEGSEECQVSREPSYAVFSTVGAFYLPLCVVLFVYWKIYKAAKFRVGSRKTNSVSPISEAVEVKDSAKQPQMVFTVRHATVTFQPEGDTWREQKEQRAALMVGILIGVFVLCWIPFFLTELISPLCSCDIPAIWKSIFLWLGYSNSFFNPLIYTAFNKNYNSAFKNFFSRQH
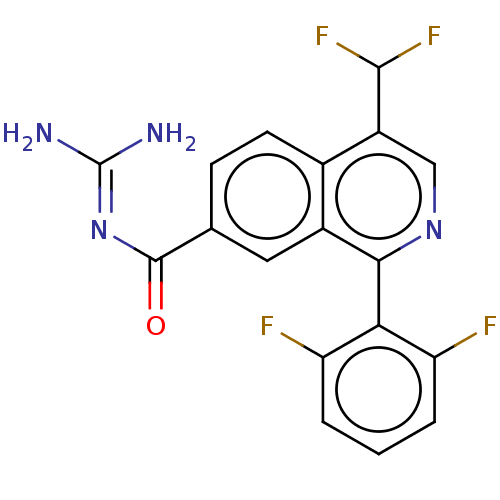
Inhibitor
Name:
BDBM135794
Synonyms:
US8853242, 187
Type:
Small organic molecule
Emp. Form.:
C18H12F4N4O
Mol. Mass.:
376.3077
SMILES:
NC(N)=NC(=O)c1ccc2c(cnc(-c3c(F)cccc3F)c2c1)C(F)F |(6,-.38,;4.67,.39,;4.67,1.93,;3.33,-.39,;2,.39,;2,1.93,;.67,-.38,;.67,-1.93,;-.67,-2.7,;-2,-1.93,;-3.33,-2.7,;-4.67,-1.93,;-4.67,-.39,;-3.33,.38,;-3.33,1.93,;-4.67,2.7,;-6,1.92,;-4.67,4.23,;-3.33,5,;-2,4.24,;-2,2.69,;-.67,1.92,;-2,-.39,;-.67,.39,;-3.33,-4.24,;-2,-5.01,;-4.67,-5.01,)|