BDBM118299 US8653092, 67
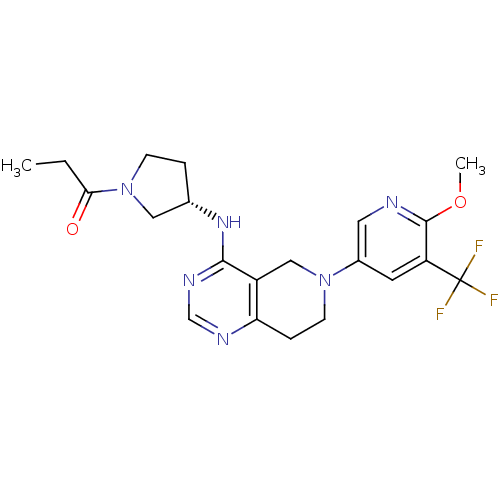
SMILES CCC(=O)N1CC[C@@H](C1)Nc2c3c(ncn2)CCN(C3)c4cc(c(nc4)OC)C(F)(F)F
InChI Key InChIKey=MWKYMZXCGYXLPL-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 40 hits for monomerid = 118299
Found 40 hits for monomerid = 118299
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 1.5nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using PIP2 as substrate measured after 1 hr in presence of ATP by ADP-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Mouse)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of PI3Kdelta in anti-CD3/CD28 stimulated mouse CD4-positive T cells assessed as reduction in T cell differentiation to Th2 cells by measur...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of PI3Kdelta (unknown origin) using phosphatidyl inositol as substrate measured after 60 mins by Alexa Fluor647-labelled ADP tracer based ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:Test of Lipid Kinase Activity: The kinase reaction is performed in a final volume of 50 μl per well of a half area COSTAR, 96 well plate. The fi...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Mouse)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 33nMAssay Description:Inhibition of PI3Kdelta in mouse spleen cells assessed as reduction in cell proliferation by measuring [3H]thymidine incorporation by beta counting b...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Mouse)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 48nMAssay Description:Inhibition of PI3Kdelta in Balb/c mouse splenocytes assessed as reduction in anti-IgM stimulated CD86 expression pretreated for 1 hr followed by anti...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Mouse)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 48nMAssay Description:Inhibition of PI3Kdelta in Balb/c mouse splenocytes assessed as reduction in anti-IgM stimulated CD86 expression pretreated for 1 hr followed by anti...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 56nMAssay Description:Inhibition of recombinant human myristoylated PI3Kdelta catalytic domain expressed in Rat1 cells assessed as reduction in Akt phosphorylation at Ser4...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 63nMAssay Description:Inhibition of PI3Kdelta in IgM stimulated human Raji cells incubated for 10 mins by AlphaLISA assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 76nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate measured after 1 hr in presence of ATP by ADP-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 79nMAssay Description:Inhibition of PI3Kdelta in human PBMC assessed as reduction in cell proliferation by measuring [3H]thymidine incorporation measured last 16 hrs of 4 ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 95nMAssay Description:Inhibition of PI3Kdelta in anti-CD3/CD28 stimulated human PBMC derived CD4-positive T cells assessed as reduction in T cell differentiation to Th2 ce...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Mouse)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 101nMAssay Description:Inhibition of PI3Kdelta in anti-CD3/CD28 stimulated mouse CD4-positive T cells assessed as reduction in T cell differentiation to Th17 cells by measu...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 144nMAssay Description:Inhibition of PI3Kdelta in human B cells assessed as reduction in anti-IgM-induced AKT phosphorylation at ser473 residue pretreated for 1.5 hrs follo...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 159nMAssay Description:Inhibition of PI3Kdelta in anti-CD3 stimulated human PBMC assessed as reduction in cell proliferation by measuring [3H]thymidine incorporation pretre...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 159nMAssay Description:Inhibition of PI3Kdelta in anti-CD3 stimulated human PBMC assessed as reduction in cell proliferation by measuring [3H]thymidine incorporation pretre...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 193nMAssay Description:Inhibition of PI3Kdelta in 90% human whole blood assessed as reduction in anti-IgM/IL4 stimulated CD69 expression pretreated for 1 hr followed by ant...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 202nMAssay Description:Inhibition of PI3Kdelta in 90% human whole blood assessed as reduction in anti-IgM/IL4 stimulated CD86 expression pretreated for 1 hr followed by ant...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 244nMAssay Description:Inhibition of recombinant human myristoylated PI3Kalpha catalytic domain expressed in Rat1 cells assessed as reduction in Akt phosphorylation at Ser4...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 262nMAssay Description:Test of Lipid Kinase Activity: The kinase reaction is performed in a final volume of 50 μl per well of a half area COSTAR, 96 well plate. The fi...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 363nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate measured after 1 hr in presence of ATP by ADP-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 424nMAssay Description:Inhibition of recombinant PI3Kbeta (unknown origin) using phosphatidyl inositol/n-Octyl-glucoside as substrate measured after 60 mins by KinaseGlo as...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 424nMAssay Description:Inhibition of recombinant human myristoylated PI3Kbeta catalytic domain expressed in Rat1 cells assessed as reduction in Akt phosphorylation at Ser47...More data for this Ligand-Target Pair
TargetDNA-dependent protein kinase catalytic subunit(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 880nMAssay Description:Inhibition of DNA-PK (unknown origin) using peptide substrate assessed as reduction in incorporation of radioactive 33p into substrate by liquid scin...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 902nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using PIP2 as substrate measured after 1 hr in presence of ATP by ADP-glo luminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 1.15E+3nMAssay Description:Inhibition of PI3Kdelta in anti-CD3/CD28 stimulated human PBMC derived CD4-positive T cells assessed as reduction in cell proliferation by measuring ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 1.67E+3nMAssay Description:Inhibition of recombinant human myristoylated PI3Kalpha catalytic domain expressed in Rat1 cells assessed as reduction in Akt phosphorylation at Ser4...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 2.23E+3nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using phosphatidyl inositol as substrate measured after 30 mins by Alexa Fluor647-labelled ADP tracer based ...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 2.68E+3nMAssay Description:Inhibition of PI3Kbeta in LPA stimulated human PC-3 cells incubated for 10 mins by AlphaLISA assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of p53 in human HCT116 cells assessed as reduction in nutlin-induced p53 transcriptional response by FRET assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Mouse)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 4.04E+3nMAssay Description:Inhibition of PI3Kgamma in C5a stimulated mouse RAW264.7 cells incubated for 10 mins by AlphaLisa assayMore data for this Ligand-Target Pair
TargetcAMP-specific 3',5'-cyclic phosphodiesterase 4D(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 4.70E+3nMAssay Description:Inhibition of recombinant human PDE4DMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Human)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 7.40E+3nMAssay Description:Inhibition of PI3Kgamma (unknown origin) using phosphatidyl inositol as substrate measured after 30 mins by Alexa Fluor647-labelled ADP tracer based ...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2B(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 7.70E+3nMAssay Description:Inhibition of 5-HT2B (unknown origin) by calcium flux assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Mouse)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 7.80E+3nMAssay Description:Inhibition of PI3Kgamma in C57/Bl6 mouse BMMC assessed as reduction in adenosine stimulated ATK phosphorylation at ser473 residue pretreated for 30 m...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 3-kinase catalytic subunit type 3(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 9.10E+3nMAssay Description:Inhibition of Vps34 (unknown origin) using phosphatidyl inositol/n-Octyl-glucoside as substrate measured after 30 mins by KinaseGlo assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 9.10E+3nMAssay Description:Inhibition of recombinant mTOR (unknown origin) using 4EBP1 as substrate by TR-FRET assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4-kinase beta(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 9.10E+3nMAssay Description:Inhibition of PI4Kbeta (unknown origin) using phosphatidyl inositol/n-Octyl-glucoside as substrate measured after 30 mins by KinaseGlo assayMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase mTOR(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of mTOR (unknown origin)More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Mouse)
Peking Union Medical College
Curated by ChEMBL
Peking Union Medical College
Curated by ChEMBL
Affinity DataIC50: 1.96E+4nMAssay Description:Inhibition of PI3Kalpha in IGF-1 stimulated mouse C2C12 cells incubated for 10 mins by AlphaLISA assayMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)