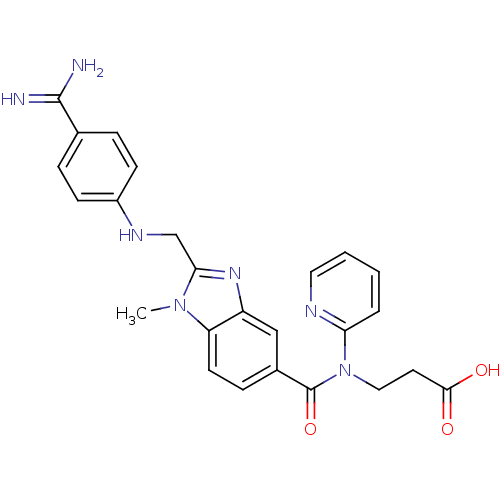
BDBM50112086 3-({2-[(4-Carbamimidoyl-phenylamino)-methyl]-1-methyl-1H-benzoimidazole-5-carbonyl}-pyridin-2-yl-amino)-propionic acid::CHEMBL48361::DABIGATRAN
SMILES [H]/N=C(c1ccc(cc1)NCc2nc3cc(ccc3n2C)C(=O)N(CCC(=O)O)c4ccccn4)/N
InChI Key InChIKey=YBSJFWOBGCMAKL-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 35 hits for monomerid = 50112086
Found 35 hits for monomerid = 50112086
Affinity DataIC50: 1.20nMAssay Description:Inhibition of human thrombin pre-incubated for 10 mins before Ac-FVR-AMC substrate addition and measured after 10 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:Inhibition of human thrombin preincubated for 10 mins followed by Ac-FVR-AMC substrate addition measured every 20s for 10 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:Inhibition of human thrombin preincubated for 10 mins followed by Ac-FVR-AMC substrate addition measured within 10 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:Inhibition of human thrombin preincubated for 10 mins followed by Ac-FVR-AMC substrate addition measured within 10 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Inhibition of human thrombin using tosyl-glycyl-prolyl-arginine-4-nitranilide acetate as substrate preincubated for 10 mins by spectrophotometer anal...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of thrombin in human blood using fluorogenic Ac-FVR-AMC as substrate by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Inhibition of human thrombin using Ac-FVR-AMC as substrate incubated for 10 mins prior to substrate addition measured for 10 min by fluorescence assa...More data for this Ligand-Target Pair
Affinity DataKi: 4.5nMAssay Description:Binding affinity to human thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 4.5nMAssay Description:Inhibitory constant (Ki) was determined against human thrombinMore data for this Ligand-Target Pair
Affinity DataKi: 4.5nMAssay Description:Inhibition of human thrombin using tosyl-glycyl-prolyl-arginine-4-nitranilide acetate as substrate preincubated for 10 mins followed by substrate add...More data for this Ligand-Target Pair
Affinity DataKi: 4.5nMAssay Description:Inhibition of human coagulation factor alpha-thrombin using Boc-Val-Pro-Arg-AMC as fluorogenic substrate measured at 1 min interval for 1 hr by fluor...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Inhibition of human thrombin assessed as reduction in release of free nitroaniline using tosyl-glycyl-prolyl-arginine-4-nitranilide acetate as substr...More data for this Ligand-Target Pair
Affinity DataKi: 6.30nMAssay Description:Inhibition of thrombin using S-2238 as substrate preincubated for 15 mins prior substrate addition measured for every 10 secs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.40nMAssay Description:Inhibition of human coagulation factor alpha-thrombin using Boc-Val-Pro-Arg-AMC as fluorogenic substrate measured at 1 min interval for 1 hr by fluor...More data for this Ligand-Target Pair
Affinity DataIC50: 9.30nMAssay Description:Inhibition of human thrombin using tosyl-glycyl-prolyl-arginine-4-nitranilide acetate as substrate preincubated for 10 mins followed by substrate add...More data for this Ligand-Target Pair
Affinity DataIC50: 9.30nMAssay Description:Inhibition of human thrombin by chromogenic assayMore data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Inhibition of human thrombin using S-2238 as chromogenic substrate preincubated for 10 mins followed by substrate addition and measured after 10 mins...More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Inhibition of human trypsinMore data for this Ligand-Target Pair
Affinity DataKi: 50.3nMAssay Description:Inhibitory constant (Ki) was determined against human trypsinMore data for this Ligand-Target Pair
Affinity DataKi: 835nMAssay Description:Inhibition of hepsin (unknown origin) using Boc-QAR-AMC as substrate after 30 mins prior to substrate addition by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Inhibition of factor-10a (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 1.70E+3nMAssay Description:Inhibitory constant (Ki) was determined against human plasminMore data for this Ligand-Target Pair
Affinity DataKi: 3.76E+3nMAssay Description:Inhibitory constant (Ki) was determined against human Coagulation factor Xa (fXa)More data for this Ligand-Target Pair
Affinity DataKi: 3.80E+3nMAssay Description:Inhibition of human coagulation factor Xa using Boc-Ile-Glu-Gly-Arg-AMC as fluorogenic substrate measured at 1 min interval for 1 hr by fluorometric ...More data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMAssay Description:Inhibition of human OCT2-mediated ASP+ uptake expressed in HEK293 cells after 3 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 5.80E+3nMAssay Description:Inhibition of human factor Xa using S-2765 as chromogenic substrate preincubated for 10 mins followed by substrate addition and measured after 10 min...More data for this Ligand-Target Pair
Affinity DataIC50: 8.10E+3nMAssay Description:Inhibition of human MATE1-mediated ASP+ uptake expressed in HEK293 cells after 1.5 mins by fluorescence assayMore data for this Ligand-Target Pair
TargetRibosyldihydronicotinamide dehydrogenase [quinone](Human)
Caprotec Bioanalytics
Curated by ChEMBL
Caprotec Bioanalytics
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Competitive inhibition of human recombinant NQO2-mediated mitomycin C metabolism using NADH as cosubstrate incubated for 5 mins prior to NADH additio...More data for this Ligand-Target Pair
TargetRibosyldihydronicotinamide dehydrogenase [quinone](Human)
Caprotec Bioanalytics
Curated by ChEMBL
Caprotec Bioanalytics
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Displacement of (S)-N4-(4-(3-(2-((4-carbamimidoylphenylamino)methyl)-1-methyl-N-(pyridin-2-yl)-1H-benzo[d]imidazole-5-carboxamido)propanamido)butyl)-...More data for this Ligand-Target Pair
Affinity DataKi: 2.09E+4nMAssay Description:Inhibitory constant (Ki) was determined against human Activated protein CMore data for this Ligand-Target Pair
Affinity DataIC50: 2.53E+4nMAssay Description:Inhibition of human MATE2K-mediated ASP+ uptake expressed in HEK293 cells after 1.5 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+4nMAssay Description:Inhibition of human coagulation factor XIIA using Boc-Gln-Gly-Arg-AMC as fluorogenic substrate measured at 1 min interval for 1 hr by fluorometric as...More data for this Ligand-Target Pair
Affinity DataKi: 4.54E+4nMAssay Description:Inhibitory constant (Ki) was determined against human Tissue plasminogen activator (tissue plasminogen activator)More data for this Ligand-Target Pair
TargetRibosyldihydronicotinamide dehydrogenase [quinone](Human)
Caprotec Bioanalytics
Curated by ChEMBL
Caprotec Bioanalytics
Curated by ChEMBL
Affinity DataKi: 6.00E+4nMAssay Description:Inhibition of human recombinant NQO2-mediated mitomycin C metabolism using NADH as cosubstrate incubated for 5 mins prior to NADH addition measured a...More data for this Ligand-Target Pair