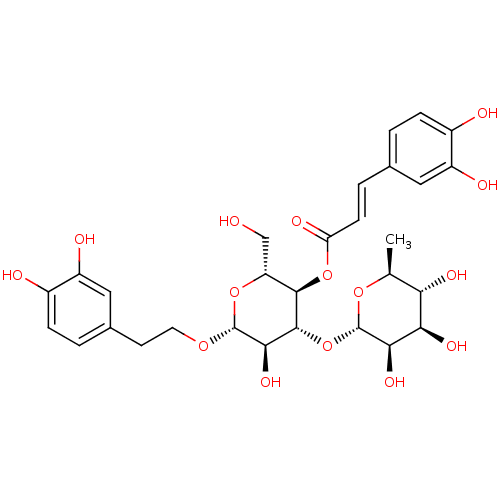
BDBM50241867 ((2R,3R,4R,5R,6R)-6-(3,4-dihydroxyphenethoxy)-5-hydroxy-2-(hydroxymethyl)-4-((2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yloxy)tetrahydro-2H-pyran-3-yl) 3-(3,4-dihydroxyphenyl)acrylate::((2R,3R,4R,5R,6R)-6-(3,4-dihydroxyphenethoxy)-5-hydroxy-2-(hydroxymethyl)-4-((2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yloxy)tetrahydro-2H-pyran-3-yl)3-(3,4-dihydroxyphenyl)acrylate::ACETOSIDE::CHEMBL231853::cid_24978601::cid_5281800::verbacoside::verbascoside
SMILES C[C@@H]1O[C@@H](O[C@@H]2[C@@H](O)[C@H](OCCc3ccc(O)c(O)c3)O[C@H](CO)[C@H]2OC(=O)\C=C\c2ccc(O)c(O)c2)[C@H](O)[C@H](O)[C@H]1O
InChI Key InChIKey=FBSKJMQYURKNSU-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50241867
Found 13 hits for monomerid = 50241867
Affinity DataIC50: 6.30nMAssay Description:Inhibition of BACE1 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:Displacement of [3H]PDBu from rat brain PKC by scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 700nMAssay Description:Competitive inhibition of Leishmania amazonensis arginase assessed as inhibition constant for enzyme-inhibitor complex using L-arginine as substrate ...More data for this Ligand-Target Pair
TargetPC4 and SFRS1-interacting protein(Human)
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataIC50: 1.41E+3nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Affiliation: Ohio State University Assay Provider: Mam...More data for this Ligand-Target Pair
TargetIntegrase(Human immunodeficiency virus type 1)
Institute of Science and Technology
Curated by ChEMBL
Institute of Science and Technology
Curated by ChEMBL
Affinity DataIC50: 7.80E+3nMAssay Description:Inhibitory activity against HIV-1 integraseMore data for this Ligand-Target Pair
Affinity DataEC50: 9.83E+3nMAssay Description:Broad Institute: MLPCN maternal gene expression Project ID: 2024 Keywords: Zinc finger, C. elegans, maternal gene expression, RNA-protein interac...More data for this Ligand-Target Pair
Affinity DataIC50: 1.13E+4nMAssay Description:Inhibition of recombinant ovine COX2 using arachidonic acid as substrate preincubated for 15 mins followed by arachidonic acid addition measured afte...More data for this Ligand-Target Pair
Affinity DataEC50: 2.06E+4nMAssay Description:Broad Institute: MLPCN maternal gene expression Project ID: 2024 Keywords: Zinc finger, C. elegans, maternal gene expression, RNA-protein interac...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of rat brain PKC catalytic domain by mixed micellar assayMore data for this Ligand-Target Pair
Affinity DataKi: 2.20E+4nMAssay Description:Displacement of [gamma32P]ATP from rat brain PKC by competitive Lineweaver-Burke plot analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of rat brain PKC by mixed micellar assayMore data for this Ligand-Target Pair
Affinity DataKi: 2.80E+4nMAssay Description:Inhibition of rat brain PKC using histone type 3-S as substrate by noncompetitive Lineweaver-Burke plot analysisMore data for this Ligand-Target Pair
TargetPC4 and SFRS1-interacting protein(Human)
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
The Scripps Research Institute Molecular Screening Center
Curated by PubChem BioAssay
Affinity DataIC50: 3.17E+4nMAssay Description:Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center (SRIMSC) Affiliation: Ohio State University Assay Provider: Mam...More data for this Ligand-Target Pair