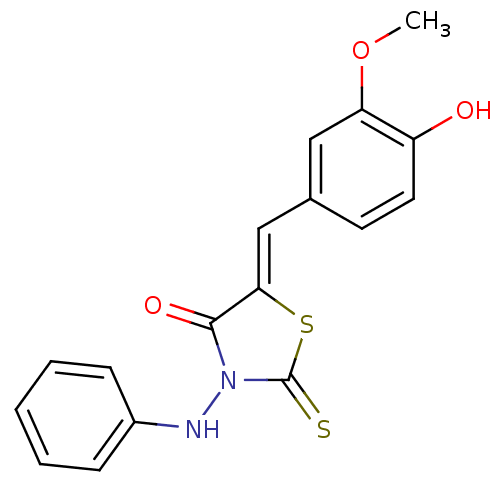
BDBM48081 (5Z)-3-anilino-2-thioxo-5-vanillylidene-thiazolidin-4-one::(5Z)-3-anilino-5-[(4-hydroxy-3-methoxyphenyl)methylidene]-2-sulfanylidene-1,3-thiazolidin-4-one::(5Z)-3-anilino-5-[(4-hydroxy-3-methoxyphenyl)methylidene]-2-sulfanylidene-4-thiazolidinone::(5Z)-5-[(3-methoxy-4-oxidanyl-phenyl)methylidene]-3-phenylazanyl-2-sulfanylidene-1,3-thiazolidin-4-one::5-(4-Hydroxy-3-methoxy-benzylidene)-3-phenylamino-2-thioxo-thiazolidin-4-one::MLS000555046::SMR000171742::cid_1202405
SMILES COc1cc(\C=C2/SC(=S)N(Nc3ccccc3)C2=O)ccc1O
InChI Key InChIKey=FDFHGZDQTBXWIJ-GDNBJRDFSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 48081
Found 2 hits for monomerid = 48081
TargetKappa-type opioid receptor(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 8.41E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetBeta-galactosidase(Escherichia coli)
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: 9.67E+3nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair