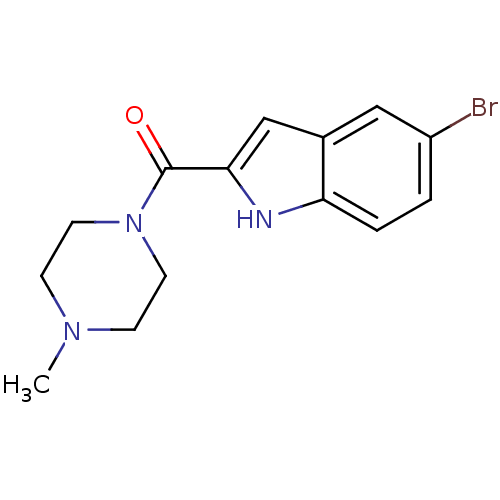
BDBM50133005 (5-Bromo-1H-indol-2-yl)-(4-methyl-piperazin-1-yl)-methanone::(5-bromo-1H-indol-2-yl)(4-methylpiperazin-1-yl)methanone::CHEMBL128613
SMILES CN1CCN(CC1)C(=O)c1cc2cc(Br)ccc2[nH]1
InChI Key InChIKey=UNZKXQGDHWYPDV-UHFFFAOYSA-N
Data 4 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50133005
Found 4 hits for monomerid = 50133005
TargetHistamine H4 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 8nMAssay Description:Displacement of [3H]histamine from recombinant human histamine H4 receptor in SK-N-MC cellsMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 8nMAssay Description:Displacement of [3H]- histamine from the recombinant human histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 7.99E+6nMAssay Description:Antagonist activity at Homo sapiens (human) histamine H4 receptorMore data for this Ligand-Target Pair
TargetHistamine H4 receptor(Homo sapiens (Human))
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research And Development
Curated by ChEMBL
Affinity DataKi: 8.00E+6nMAssay Description:Antagonist activity at Homo sapiens (human) histamine H4 receptorMore data for this Ligand-Target Pair