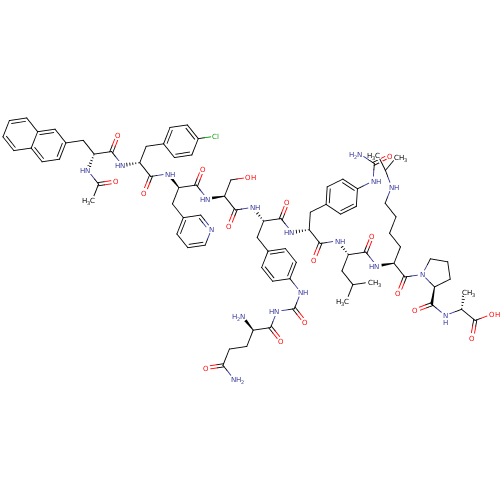
BDBM50167751 Azaline B derivative::CHEMBL413216
SMILES CC(C)C[C@H](NC(=O)[C@@H](Cc1ccc(NC(N)=O)cc1)NC(=O)[C@H](Cc1ccc(NC(=O)NC(=O)[C@H](N)CCC(N)=O)cc1)NC(=O)[C@H](CO)NC(=O)[C@@H](Cc1cccnc1)NC(=O)[C@@H](Cc1ccc(Cl)cc1)NC(=O)[C@@H](Cc1ccc2ccccc2c1)NC(C)=O)C(=O)N[C@@H](CCCCNC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@H](C)C(O)=O
InChI Key InChIKey=DJWITMRFQYLBIP-JRBVYZFBSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50167751
Found 2 hits for monomerid = 50167751
Affinity DataIC50: 6.30nMAssay Description:In vitro inhibition of human gonadotropin-releasing hormone expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 6.86nMAssay Description:In vitro inhibition of human gonadotropin-releasing hormone expressed in HEK-293 cellsMore data for this Ligand-Target Pair