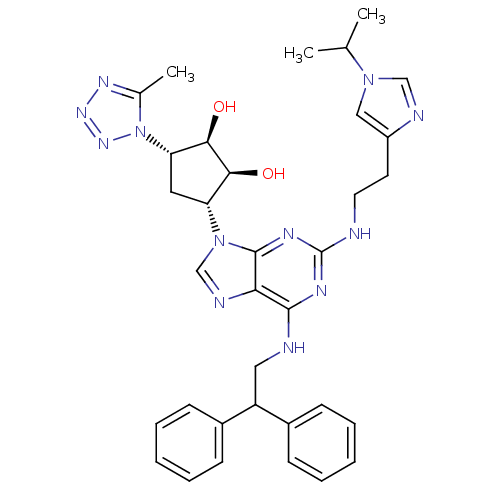
BDBM50309492 (2R,3R,4S,5S)-2-(6-(2,2-diphenylethylamino)-2-(2-(1-isopropyl-1H-imidazol-4-yl)ethylamino)-9H-purin-9-yl)-5-(5-methyl-1H-tetrazol-1-yl)tetrahydrofuran-3,4-diol::CHEMBL601732
SMILES CC(C)n1cnc(CCNc2nc(NCC(c3ccccc3)c3ccccc3)c3ncn([C@@H]4C[C@@H]([C@@H](O)[C@H]4O)n4nnnc4C)c3n2)c1
InChI Key InChIKey=IMXHSAQVSOGNKP-HNRHMXCFSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50309492
Found 2 hits for monomerid = 50309492
TargetAdenosine receptor A2a(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataKi: 38nMAssay Description:Displacement of [3H]NECA from human recombinant adenosine A2A receptor expressed in Sf21 cells co-expressing GalphaS2, beta4, gamma2More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 101nMAssay Description:Agonist activity at adenosine A2A receptor in human neutrophils assessed as inhibition of fMLP-induced reactive oxygen species release by chemilumine...More data for this Ligand-Target Pair