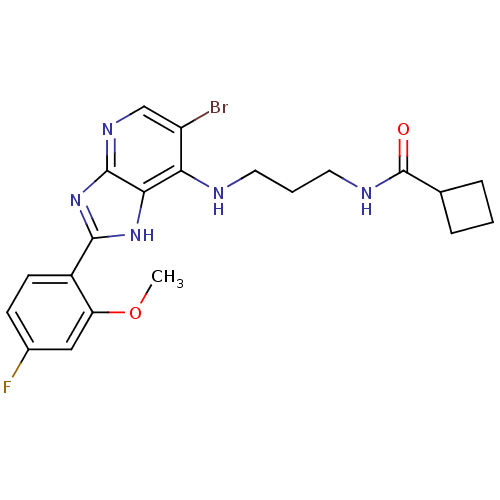
BDBM50379368 CHEMBL2010815
SMILES COc1cc(F)ccc1-c1nc2ncc(Br)c(NCCCNC(=O)C3CCC3)c2[nH]1
InChI Key InChIKey=RYGAYBGVUQJRJA-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50379368
Found 5 hits for monomerid = 50379368
Affinity DataIC50: 664nMAssay Description:Inhibition of Aurora B kinaseMore data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase TBK1(Homo sapiens (Human))
Astrazeneca R&D Boston
Curated by ChEMBL
Astrazeneca R&D Boston
Curated by ChEMBL
Affinity DataIC50: 38nMAssay Description:Inhibition of recombinant TBK1 using 5FAM-AhxKRRAL(ps)VASLPGL as substrate by microfluidic mobility shift assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2.02E+3nMAssay Description:Inhibition of CDK2More data for this Ligand-Target Pair
TargetSerine/threonine-protein kinase TBK1(Homo sapiens (Human))
Astrazeneca R&D Boston
Curated by ChEMBL
Astrazeneca R&D Boston
Curated by ChEMBL
Affinity DataEC50: 34nMAssay Description:Inhibition of TBK1-mediated NFkappaB activation expressed in ds-RNA activated HEK293 cells coexpressing TLR3 after 4.5 hrs by luciferase reporter gen...More data for this Ligand-Target Pair
TargetInhibitor of nuclear factor kappa-B kinase subunit epsilon(Homo sapiens (Human))
Astrazeneca R&D Boston
Curated by ChEMBL
Astrazeneca R&D Boston
Curated by ChEMBL
Affinity DataIC50: 204nMAssay Description:Inhibition of Ikkepsilon using 5FAM-AKELDQGSLCTpSFVGTLQ-NH2 as substrate by microfluidic mobility shift assayMore data for this Ligand-Target Pair