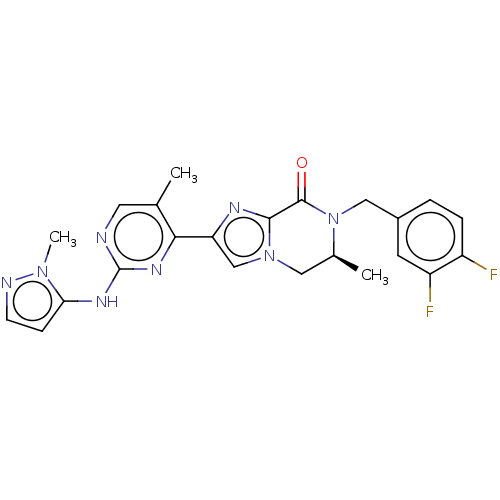
BDBM50505994 CHEMBL4440109
SMILES C[C@H]1Cn2cc(nc2C(=O)N1Cc1ccc(F)c(F)c1)-c1nc(Nc2ccnn2C)ncc1C
InChI Key InChIKey=FCBRBTSGPCWMSK-AWEZNQCLSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50505994
Found 5 hits for monomerid = 50505994
Affinity DataIC50: <0.300nMAssay Description:Inhibition of MEK U911-activated ERK2 (unknown origin) using ERKtide as substrate preincubated for 20 mins followed by substrate addition in presence...More data for this Ligand-Target Pair
Affinity DataIC50: 0.320nMAssay Description:Inhibition of MEK U911-activated ERK2 (unknown origin) using ERKtide as substrate preincubated for 20 mins followed by substrate addition in presence...More data for this Ligand-Target Pair
Affinity DataIC50: 3.40nMAssay Description:Inhibition of ERK1/2 in human A375 cells assessed as inhibition of ERK phosphorylation after 2 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:Inhibition of ERK1/2 in human A375 cells assessed as inhibition of RSK phosphorylation after 2 hrsMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 2.30E+4nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair