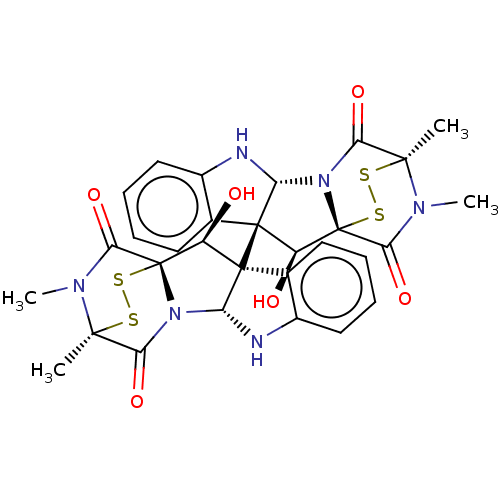
BDBM50530867 CHEMBL4472654
SMILES [H][C@]12Nc3ccccc3[C@]1([C@H](O)[C@]13SS[C@](C)(N(C)C1=O)C(=O)N23)[C@]12[C@H](O)[C@]34SS[C@](C)(N(C)C3=O)C(=O)N4[C@@]1([H])Nc1ccccc21
InChI Key InChIKey=IMGTYEJTVRXGLW-LRESJZTJSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50530867
Found 2 hits for monomerid = 50530867
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
University Of North Carolina At Greensboro
Curated by ChEMBL
University Of North Carolina At Greensboro
Curated by ChEMBL
Affinity DataIC50: 800nMAssay Description:Inhibition of human recombinant MLL1 using [3H]S-adenosyl-methionine as substrateMore data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase 2A(Homo sapiens (Human))
University Of North Carolina At Greensboro
Curated by ChEMBL
University Of North Carolina At Greensboro
Curated by ChEMBL
Affinity DataIC50: 800nMAssay Description:Inhibition of human recombinant MLL1 using [3H]S-adenosyl-methionine as substrateMore data for this Ligand-Target Pair