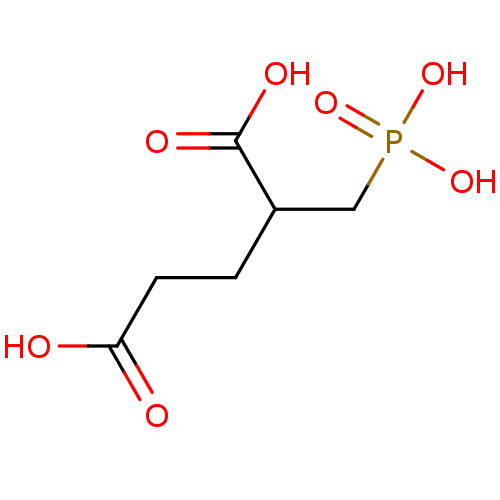
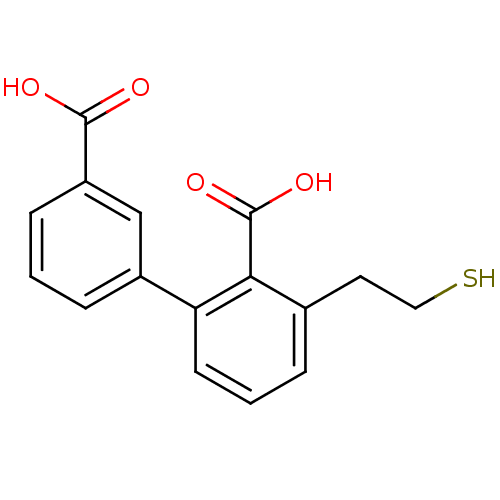
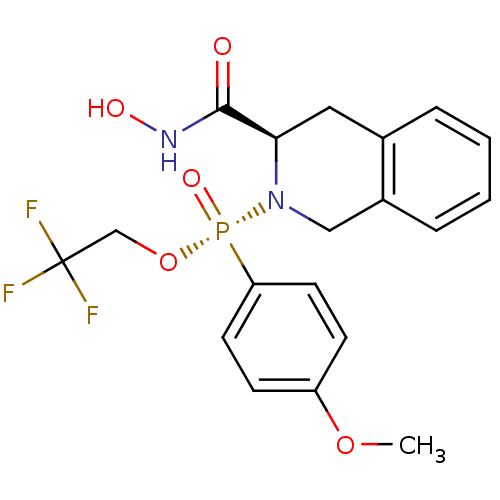
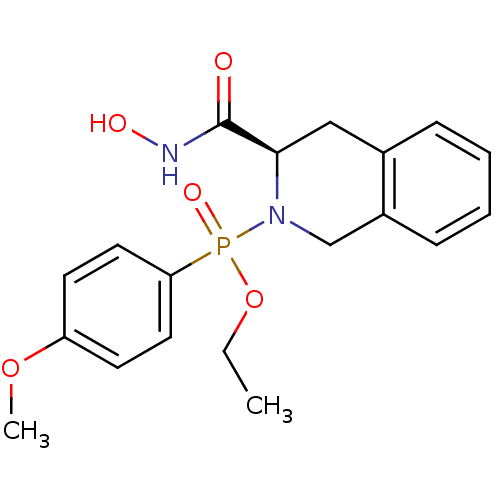
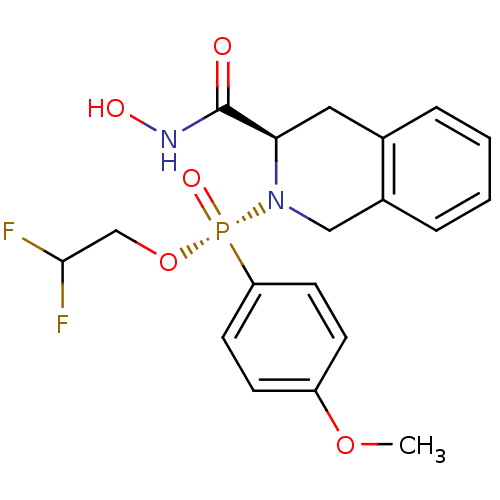
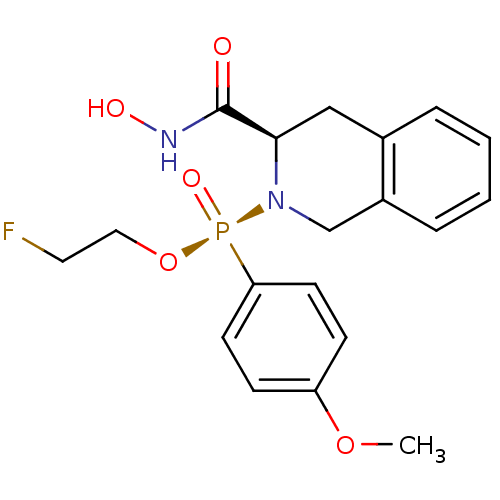
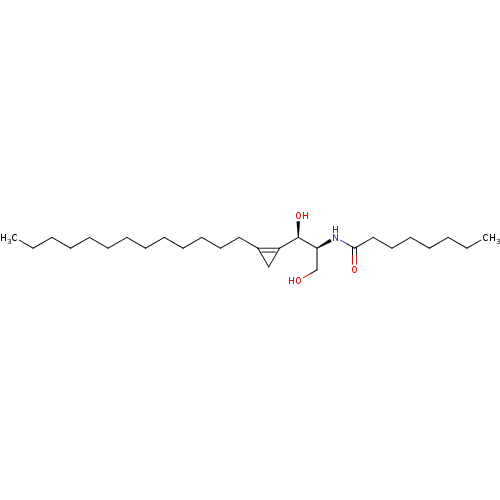
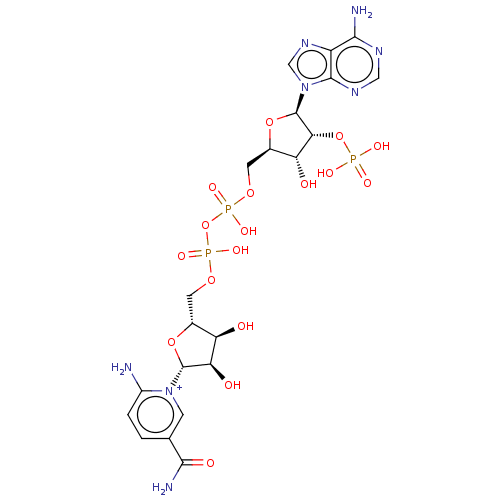
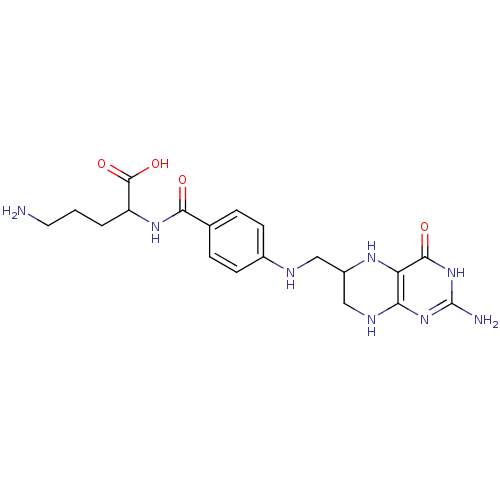
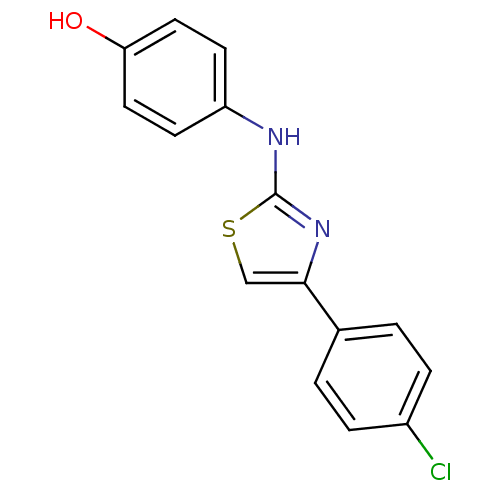
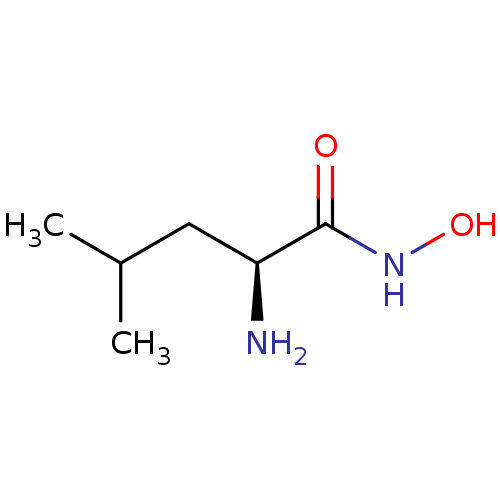
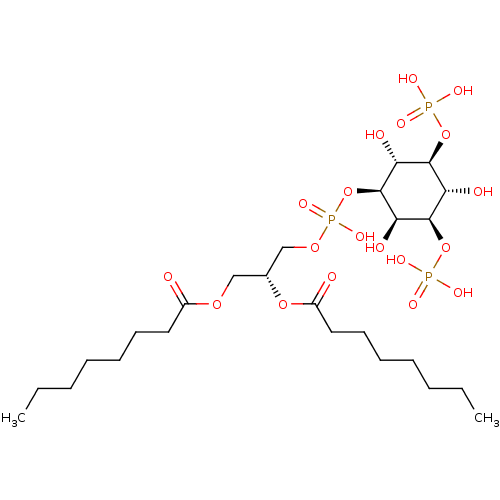
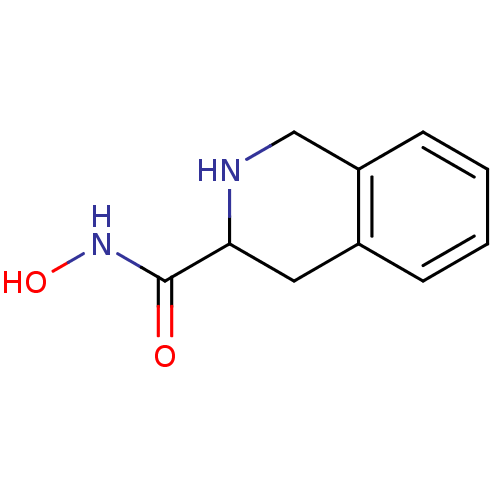
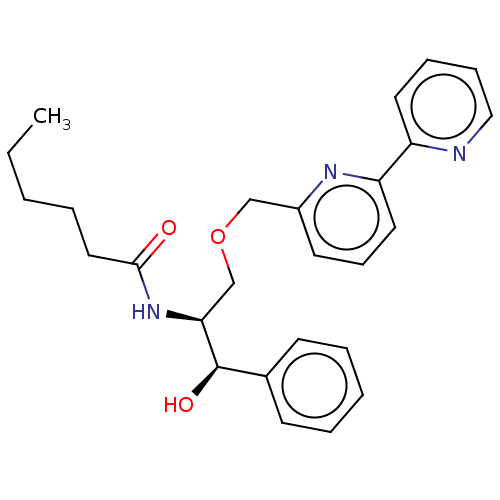
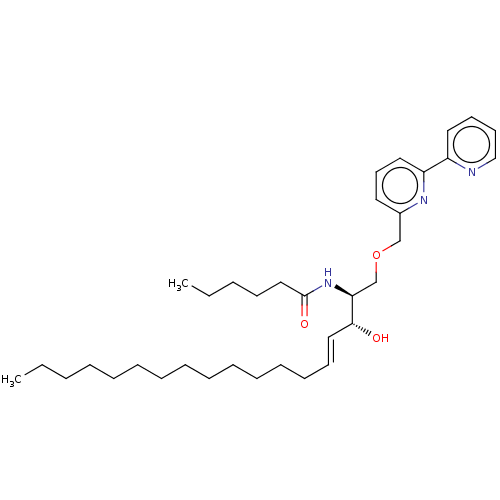
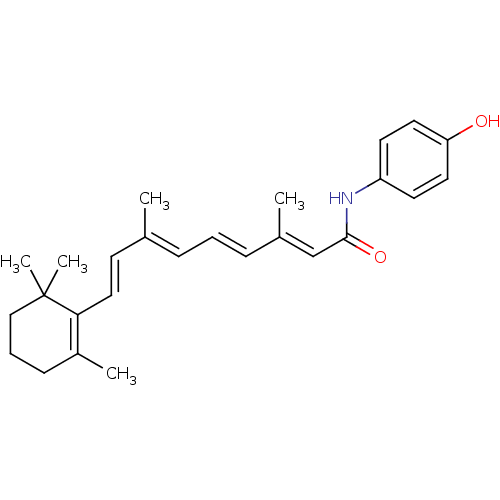
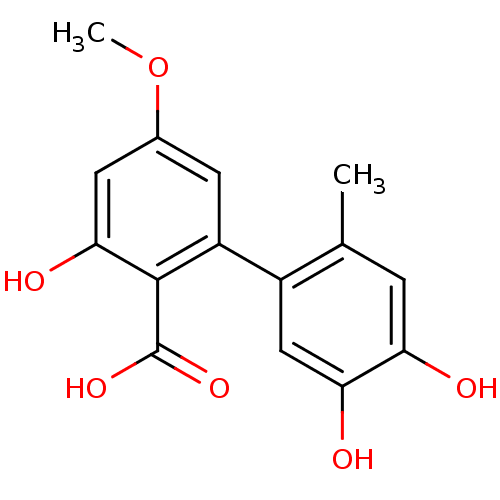
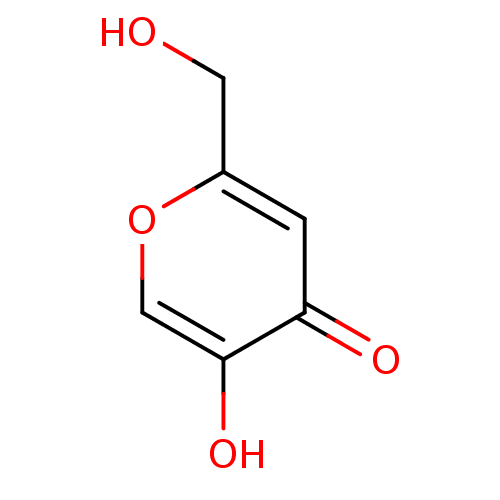
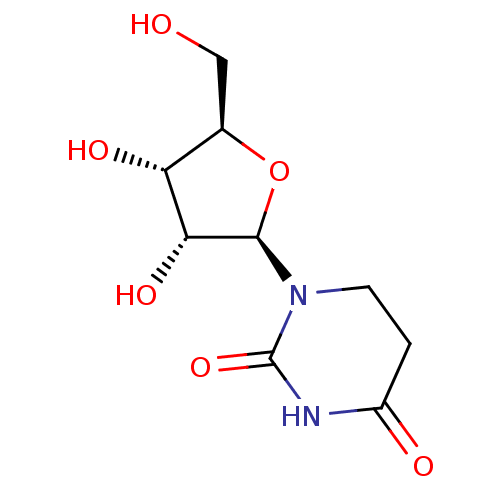
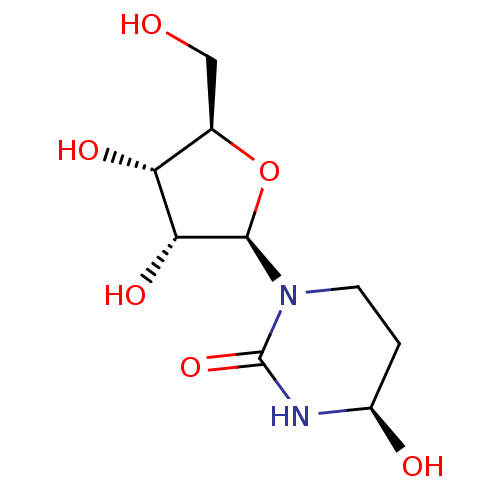
Affinity DataKi: 0.0100nMAssay Description:Inhibition of GCP2 by top scintillation counter in presence of 30 nM NAA[3]GMore data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:Concentration of the compound required for the neuroprotective effect determined by inhibition of GCP IIMore data for this Ligand-Target Pair
Affinity DataKi: 0.200nMAssay Description:In vitro inhibitory activity against glutamate carboxypeptidase II (GCP II) using N-acetyl-L-aspartyl-[3H]-L-glutamate as a substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Inhibition of GCP-2 (unknown origin)More data for this Ligand-Target Pair
TargetBacterial leucyl aminopeptidase(Vibrio proteolyticus)
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 2nMAssay Description:Inhibition of metalloprotease from family M28, Aeromonas proteolytica aminopeptidaseMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 3.30nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: 4.5nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: 4.60nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: 5.10nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: 5.20nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: 6nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 6.60nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 6.70nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: 6.80nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: 9.10nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:In vitro inhibitory activity against glutamate carboxypeptidase II (GCP II) using N-acetyl-L-aspartyl-[3H]-L-glutamate as a substrateMore data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: 22nMAssay Description:Negative log concentration of antagonist was determined on 5-hydroxytryptamine 2B receptor of Rat stomach fundusMore data for this Ligand-Target Pair
Affinity DataKi: 60nMAssay Description:Competitive inhibition of recombinant human DAAO expressed in HEK cells by double reciprocal plot analysis in presence of D-serineMore data for this Ligand-Target Pair
Target6-phosphogluconate dehydrogenase, decarboxylating(Rattus norvegicus)
Johns Hopkins University School Of Medicine
Curated by ChEMBL
Johns Hopkins University School Of Medicine
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Inhibition of rat 6PGD assessed as inhibition constant by spectrophotometric methodMore data for this Ligand-Target Pair
TargetFolylpolyglutamate synthase, mitochondrial(Homo sapiens (Human))
University of Michigan
Curated by ChEMBL
University of Michigan
Curated by ChEMBL
Affinity DataKi: 200nMAssay Description:Inhibitory activity against mammalian Folyl-polyglutamate synthaseMore data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Negative log concentration of antagonist on 5-hydroxytryptamine 2A receptor in rat thoracic aortaMore data for this Ligand-Target Pair
TargetBacterial leucyl aminopeptidase(Vibrio proteolyticus)
Guilford Pharmaceuticals
Curated by ChEMBL
Guilford Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 350nMAssay Description:Inhibition of metalloprotease from family M28, Aeromonas proteolytica aminopeptidaseMore data for this Ligand-Target Pair
Affinity DataKi: 530nMAssay Description:Negative log concentration of antagonistic compound was determined on 5-hydroxytryptamine 2B receptor of Rat stomach fundusMore data for this Ligand-Target Pair
Affinity DataKi: >650nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: >650nMAssay Description:Inhibition of Matrix metalloprotease-3 (MMP-3)More data for this Ligand-Target Pair
Affinity DataKi: >800nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: >800nMAssay Description:Inhibition of Matrix metalloprotease-9 (MMP-9)More data for this Ligand-Target Pair
Affinity DataKi: >850nMAssay Description:Inhibition of Matrix metalloprotease-1 (MMP-1)More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+3nMAssay Description:Functional potency against Tachykinin receptor 2 (NK2) in rabbit pulmonary artery assayMore data for this Ligand-Target Pair
TargetGlutaminase kidney isoform, mitochondrial(Homo sapiens (Human))
Johns Hopkins University
Curated by ChEMBL
Johns Hopkins University
Curated by ChEMBL
Affinity DataKi: 2.00E+3nMAssay Description:Uncompetitive inhibition of human kidney glutaminase (124 to 669) assessed as reduction of glutamine hydrolysis by double-reciprocal plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 2.15E+3nMAssay Description:Negative log concentration of antagonist was determined on 5-hydroxytryptamine 2B receptor of Rat stomach fundusMore data for this Ligand-Target Pair
Affinity DataKi: 2.80E+3nMAssay Description:Functional potency against Tachykinin receptor 2 (NK2) in rabbit pulmonary artery assayMore data for this Ligand-Target Pair
Affinity DataKi: 5.20E+3nMAssay Description:Functional potency against Tachykinin receptor 2 (NK2) in rabbit pulmonary artery assayMore data for this Ligand-Target Pair
Affinity DataKi: 7.00E+3nMAssay Description:Functional potency against Tachykinin receptor 2 (NK2) in rabbit pulmonary artery assayMore data for this Ligand-Target Pair
Affinity DataKi: 7.90E+3nMAssay Description:Negative log concentration of antagonist on 5-hydroxytryptamine 2A receptor in rat thoracic aortaMore data for this Ligand-Target Pair
Affinity DataKi: 8.28E+3nMAssay Description:Negative log concentration of antagonist on 5-hydroxytryptamine 2A receptor in rat thoracic aortaMore data for this Ligand-Target Pair
Affinity DataKi: 1.60E+4nMAssay Description:Negative log concentration of antagonist was determined on 5-hydroxytryptamine 2B receptor of Rat stomach fundusMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+4nMAssay Description:Negative log concentration of antagonist was determined on 5-hydroxytryptamine 2B receptor of Rat stomach fundusMore data for this Ligand-Target Pair
Affinity DataKi: 2.10E+4nMAssay Description:Competitive inhibition of pig kidney DAAO using D-Alanine as substrate by Michaelis-Menten plot analysisMore data for this Ligand-Target Pair
Affinity DataKi: 4.00E+4nMAssay Description:Inhibition of human cytidine deaminase by spectrophotometricallyMore data for this Ligand-Target Pair
Affinity DataKi: 7.27E+4nMAssay Description:Displacement of [3H]DAMGO from mu opioid receptor in rat brain after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 2.20E+5nMAssay Description:Displacement of [3H]DADLE from delta opioid receptor in rat brain after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 4.40E+5nMAssay Description:Inhibition of human cytidine deaminase by spectrophotometricallyMore data for this Ligand-Target Pair
Affinity DataKi: 1.43E+6nMAssay Description:Displacement of [3H]U-69,593 from kappa opioid receptor in rat brain after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 2.47E+6nMAssay Description:Displacement of [3H]DADLE from delta opioid receptor in rat brain after 60 mins by liquid scintillation counting methodMore data for this Ligand-Target Pair

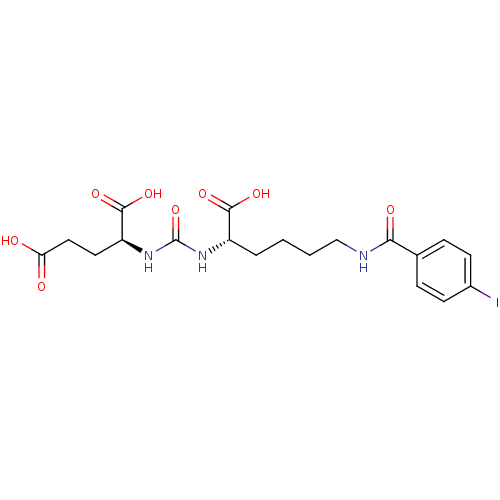
 3D Structure (crystal)
3D Structure (crystal)