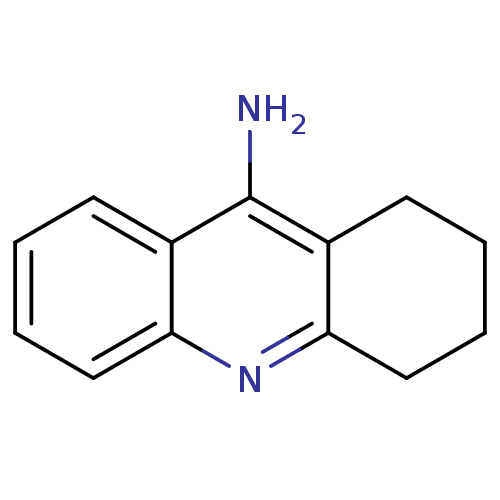
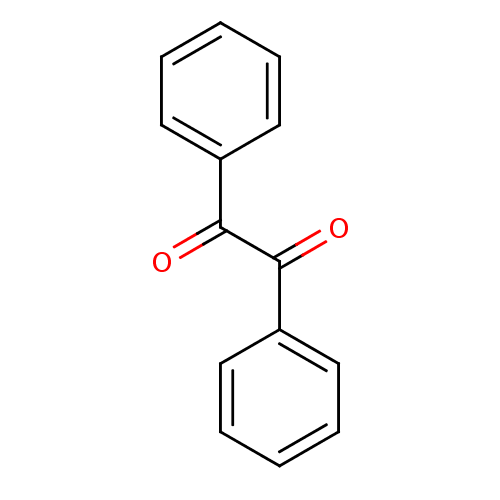
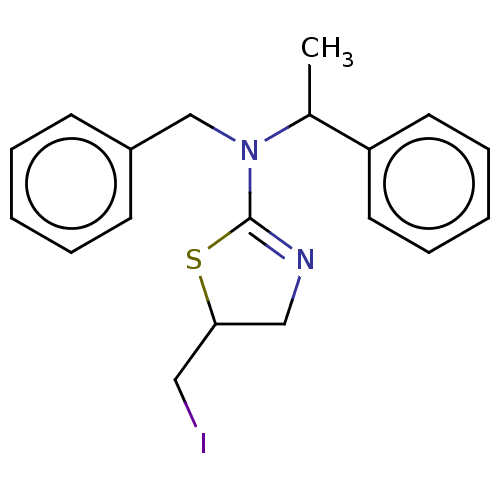
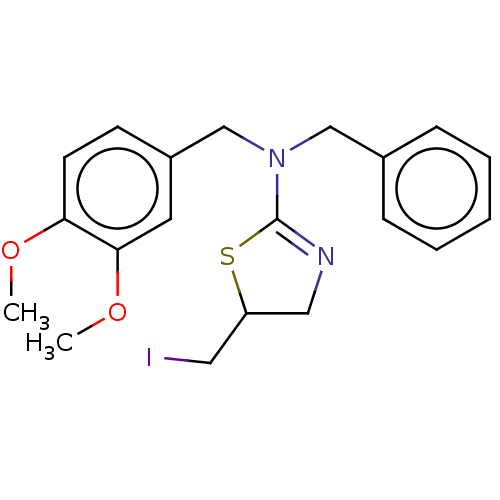
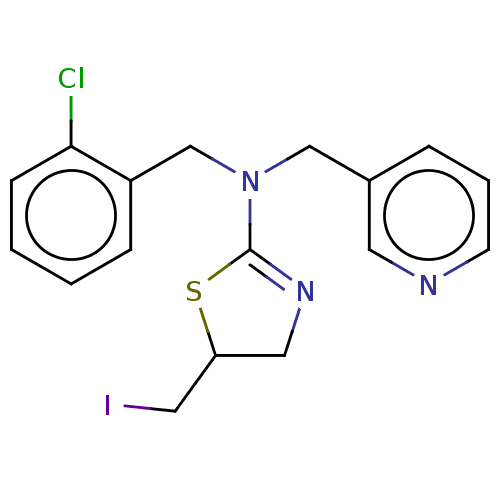
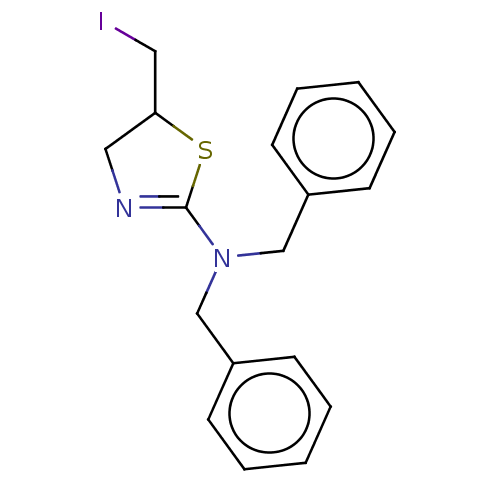
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
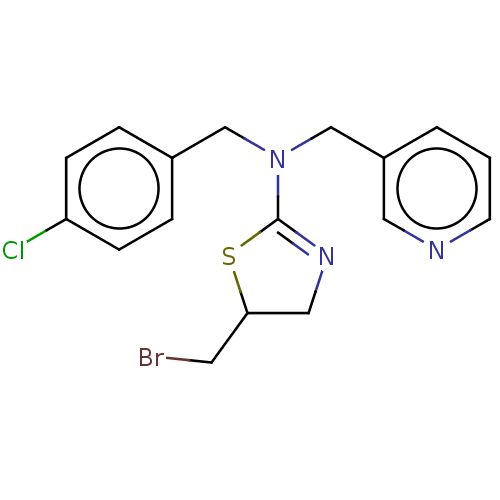
Affinity DataKi: 270nMAssay Description:Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated fo...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
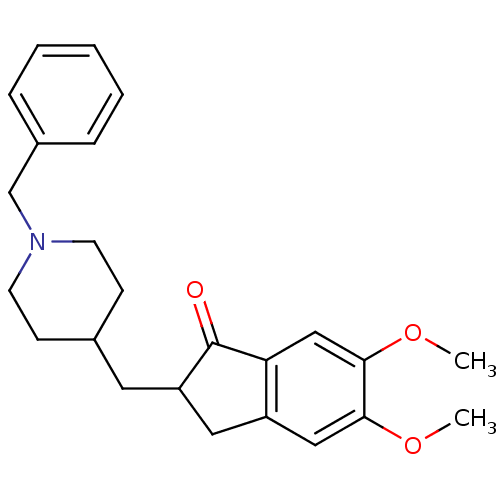
Affinity DataKi: 400nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 440nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 620nMAssay Description:Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated fo...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 670nMAssay Description:Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated fo...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 1.20E+3nMAssay Description:Noncompetitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 1.34E+3nMAssay Description:Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated fo...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 1.53E+3nMAssay Description:Noncompetitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 2.09E+3nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 3.54E+3nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 3.60E+3nMAssay Description:Noncompetitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 7.37E+3nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 7.56E+3nMAssay Description:Noncompetitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 7.81E+3nMAssay Description:Noncompetitive inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate assessed as steady state inhibition constant pre...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 9.51E+3nMAssay Description:Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated fo...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataKi: 1.99E+4nMAssay Description:Noncompetitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate assessed as steady state inhibition constant preincubated...More data for this Ligand-Target Pair
TargetAcetylcholinesterase(Homo sapiens (Human))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 22nMAssay Description:Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's ...More data for this Ligand-Target Pair
TargetCholinesterase(Homo sapiens (Human))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 34nMAssay Description:Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated followed by substrate addition by Ellman's methodMore data for this Ligand-Target Pair
TargetAcetylcholinesterase(Homo sapiens (Human))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 79nMAssay Description:Inhibition of human erythrocyte AChE using acetylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's ...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 220nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 320nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetLiver carboxylesterase 1(Homo sapiens (Human))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 370nMAssay Description:Inhibition of human carboxylesterase 1 using o-nitrophenylacetate as substrate by spectrophotometry analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 770nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.13E+3nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.34E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.47E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.55E+3nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.74E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 4.14E+3nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 4.66E+3nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Homo sapiens (Human))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 7.60E+3nMAssay Description:Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated followed by substrate addition by Ellman's methodMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.40E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.70E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 1.90E+4nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.60E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 2.90E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 3.20E+4nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetCholinesterase(Equus caballus (Horse))
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 3.80E+4nMAssay Description:Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 10 mins followed by substrate addition by Ellman's meth...More data for this Ligand-Target Pair
TargetLiver carboxylesterase(Sus scrofa)
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Institute Of Physiologically Active Compounds Russian Academy Of Sciences
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of porcine liver carboxylesterase using 4-nitrophenol acetate as substrate by spectrophotometric analysisMore data for this Ligand-Target Pair

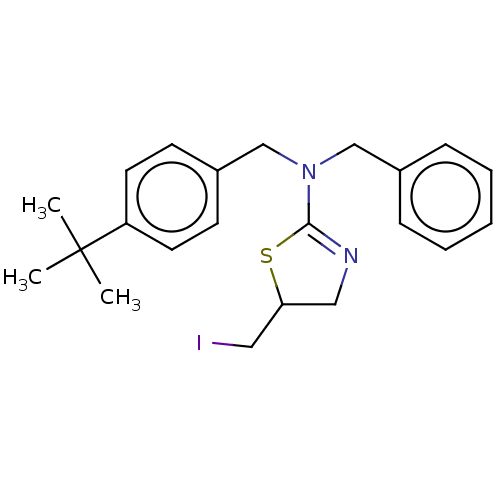
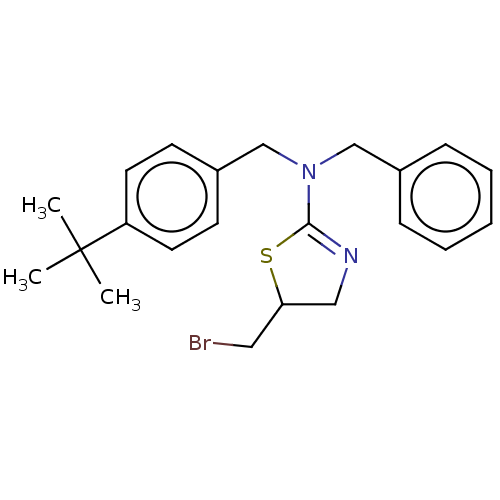






 3D Structure (crystal)
3D Structure (crystal)