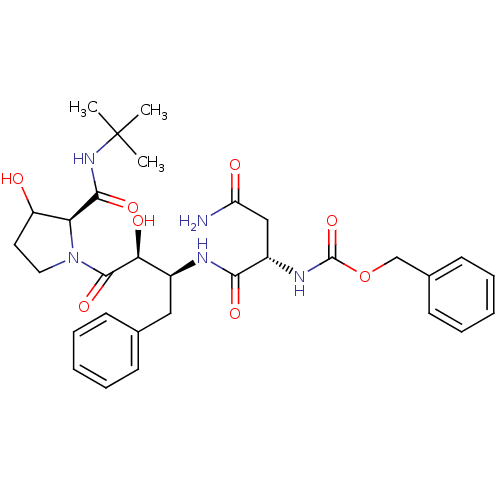
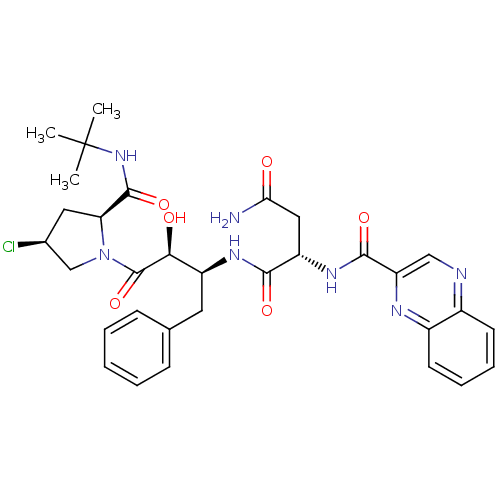
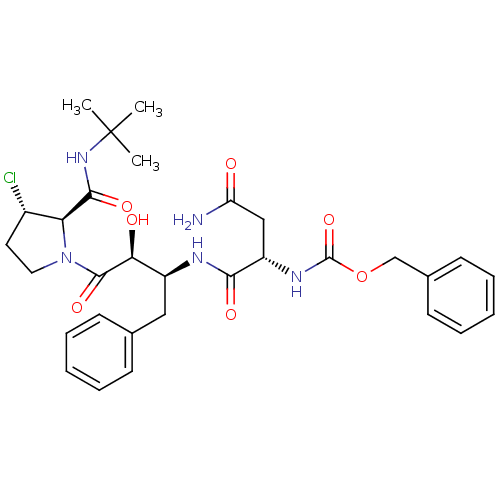
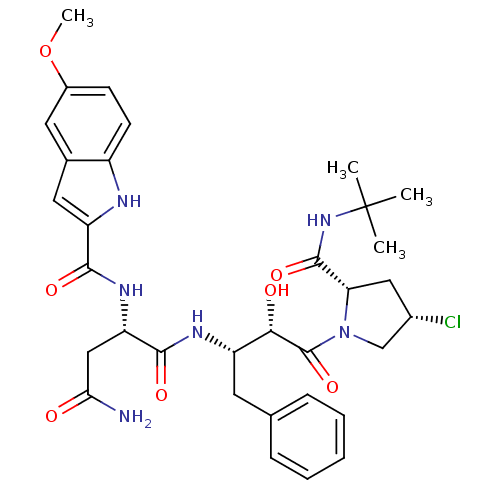
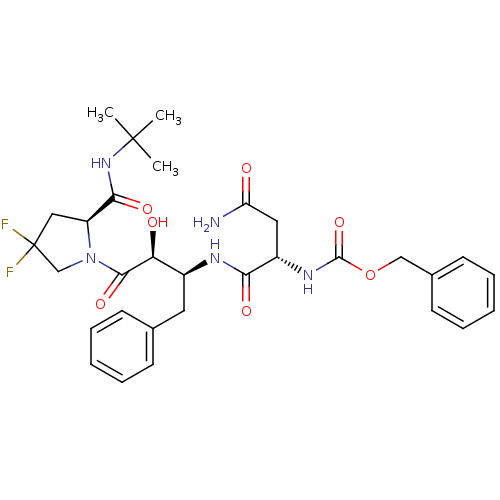
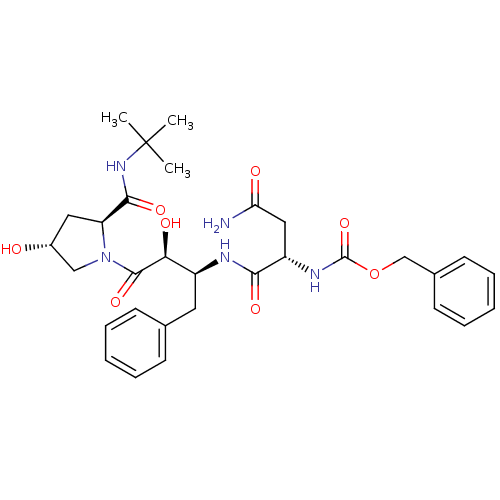
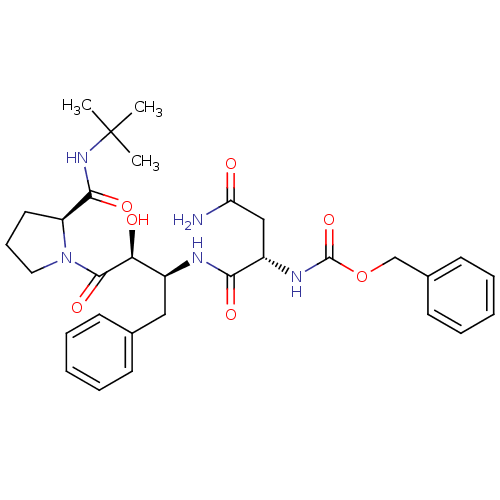
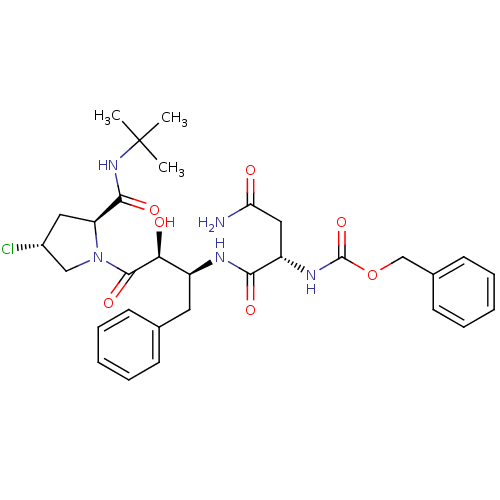
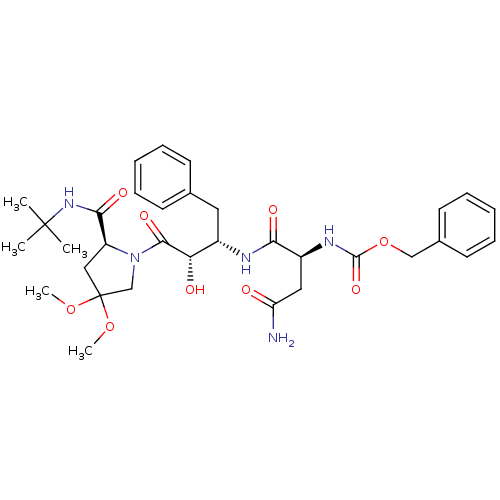
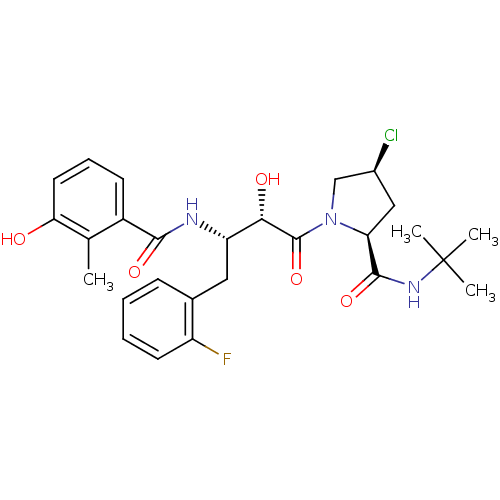
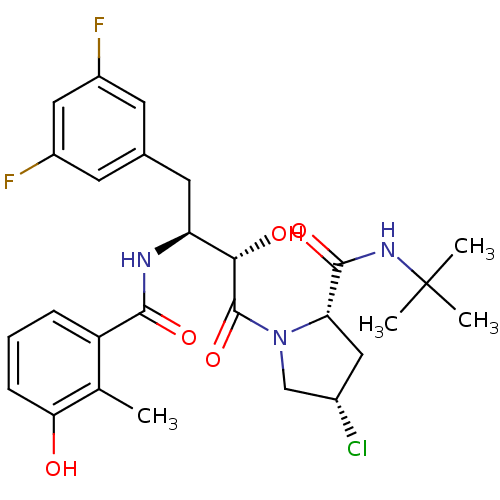
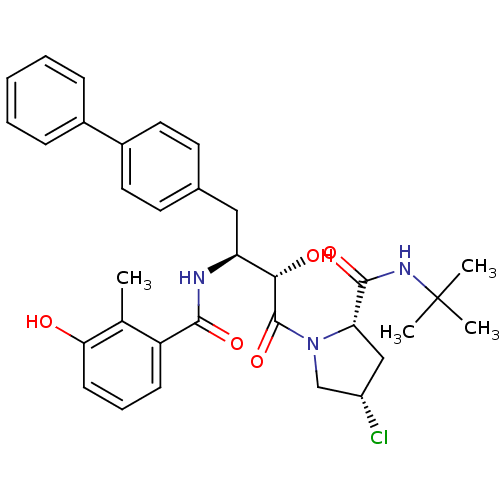
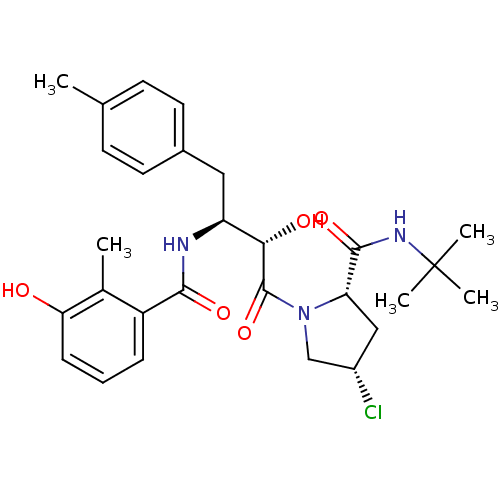
Affinity DataKi: 0.100nM ΔG°: -59.4kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
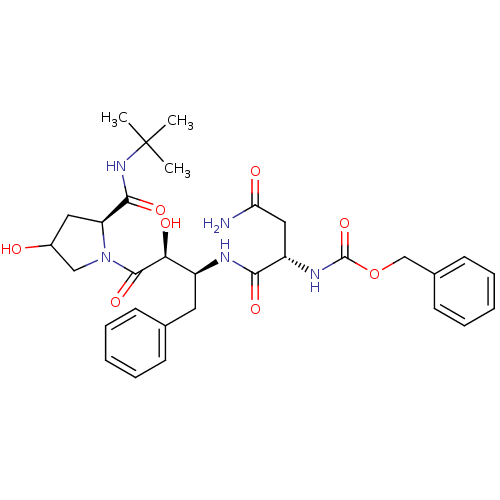
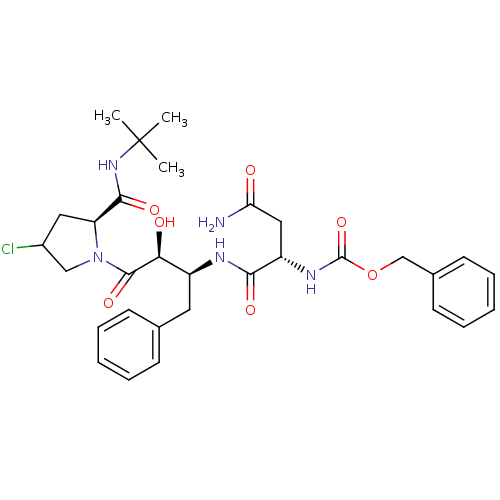
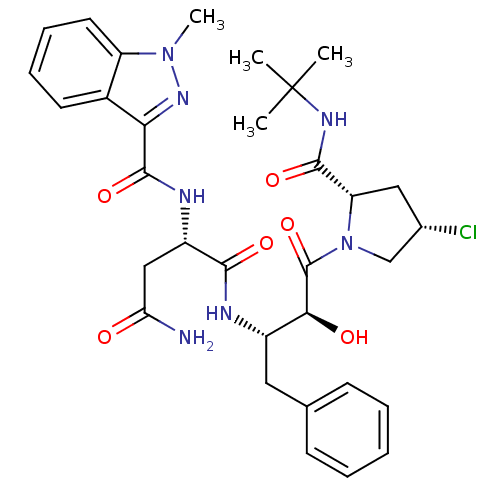
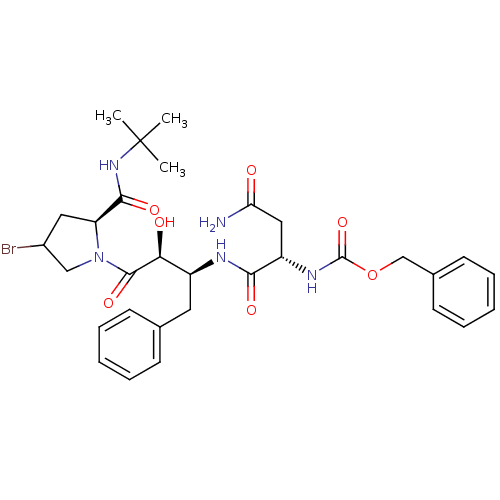
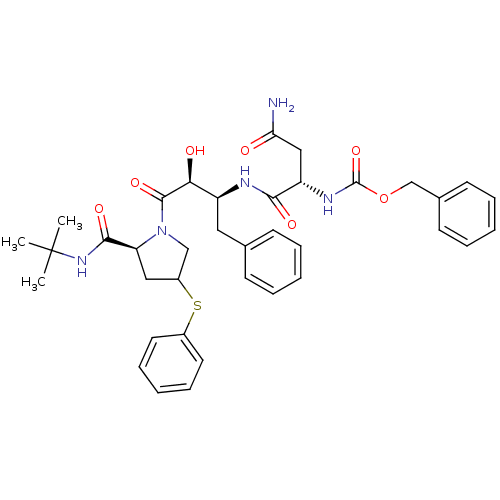
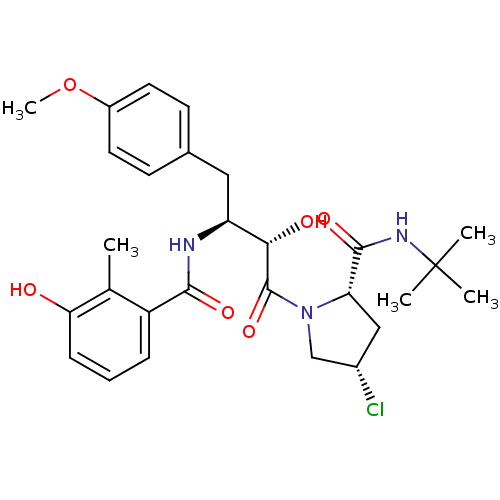
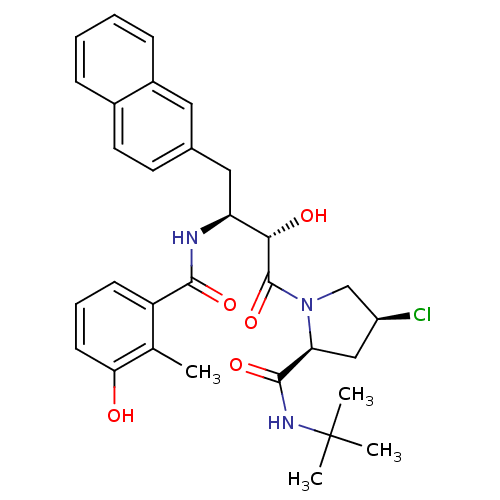
Affinity DataKi: 1nM ΔG°: -53.4kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
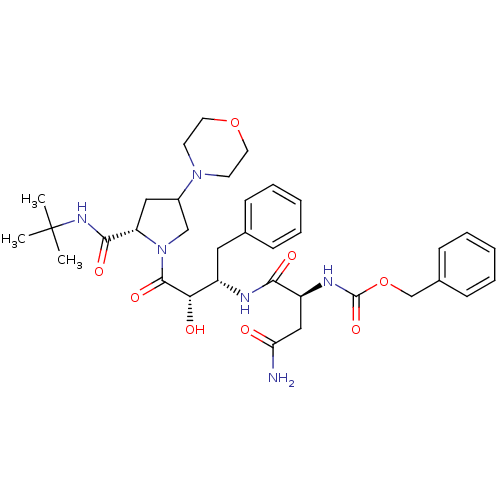
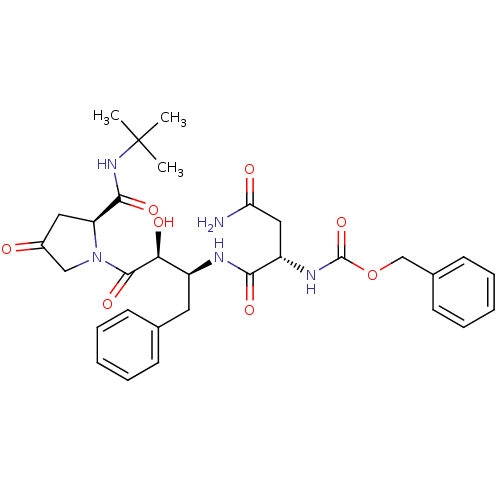
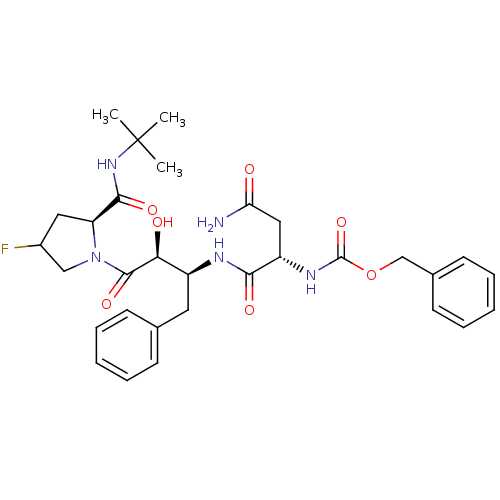
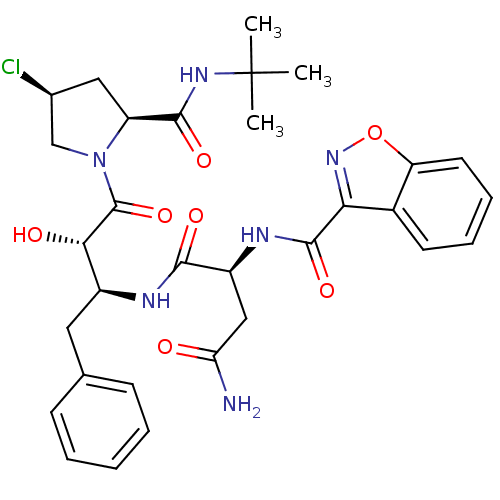
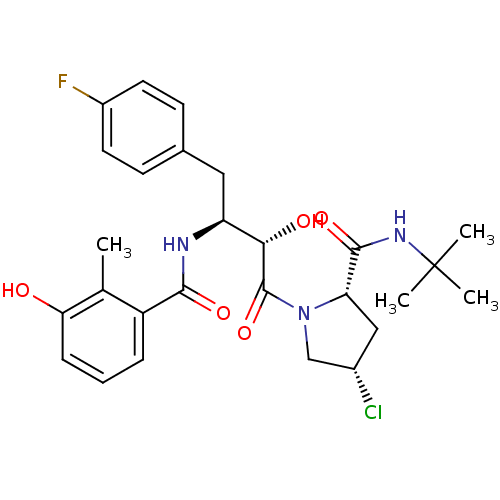
Affinity DataKi: 1nM ΔG°: -53.4kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
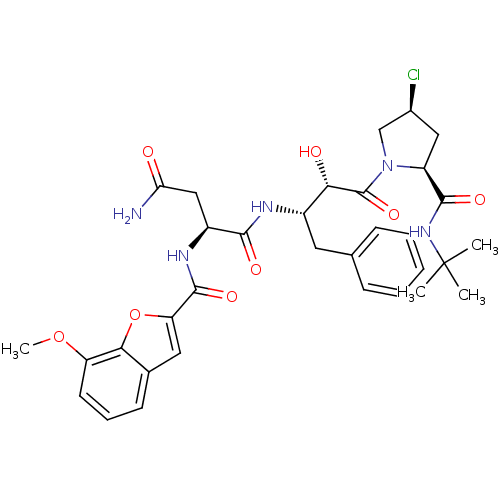
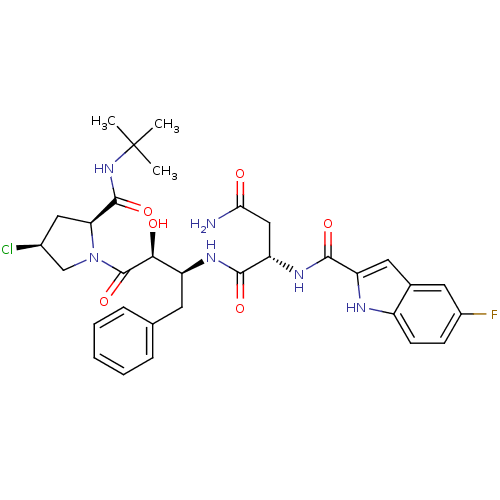
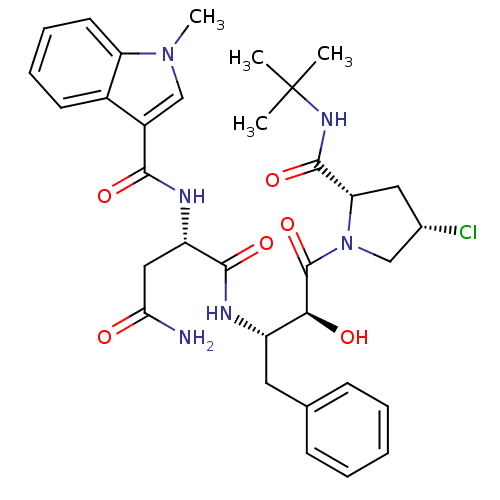
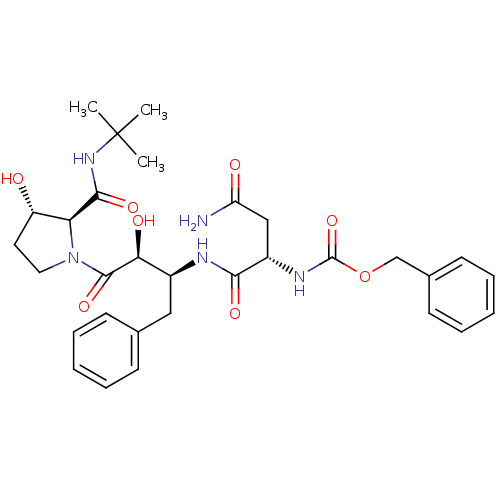
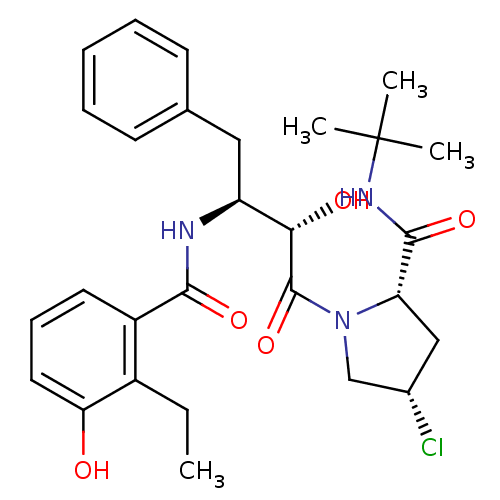
Affinity DataKi: 4.5nM ΔG°: -49.6kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 4.70nM ΔG°: -49.4kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 8nM ΔG°: -48.1kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 12.5nM ΔG°: -46.9kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 12.5nM ΔG°: -46.9kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 14.5nM ΔG°: -46.5kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 16nM ΔG°: -46.3kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 18nM ΔG°: -46.0kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 20nM ΔG°: -45.7kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 21.5nM ΔG°: -45.5kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 22.5nM ΔG°: -45.4kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 28nM ΔG°: -44.8kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 32nM ΔG°: -44.5kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 35nM ΔG°: -44.3kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 56nM ΔG°: -43.1kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 57.5nM ΔG°: -43.0kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 90nM ΔG°: -41.8kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 92nM ΔG°: -41.8kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataKi: 125nM ΔG°: -41.0kJ/molepH: 4.7 T: 2°CAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
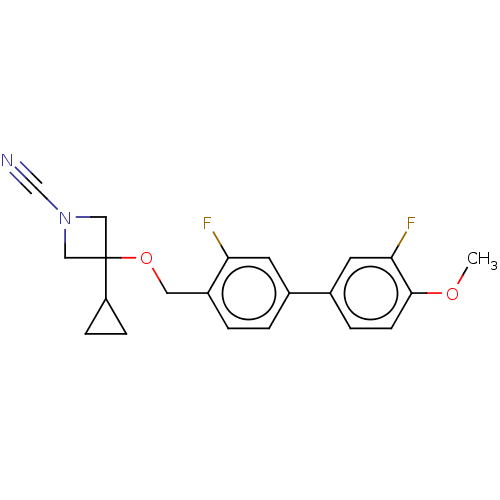
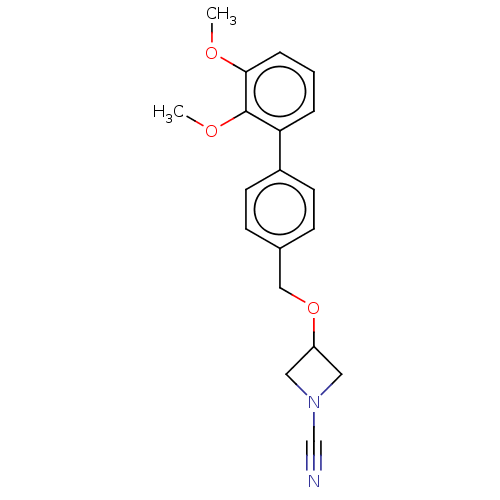
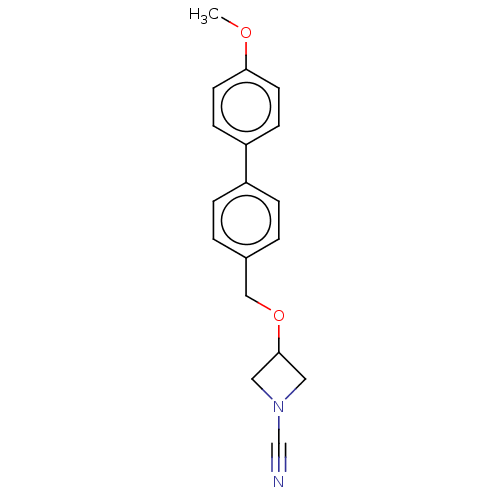
Affinity DataIC50: 0.350nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:Inhibition of recombinant human FAAH expressed in Escherichia coli using fluorogenic AAMCA as substrate by fluorimetric assayMore data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
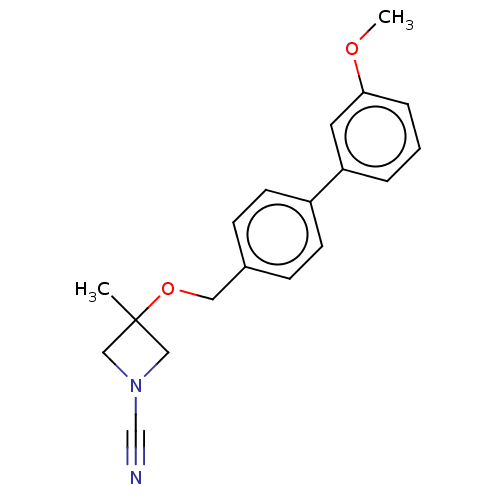
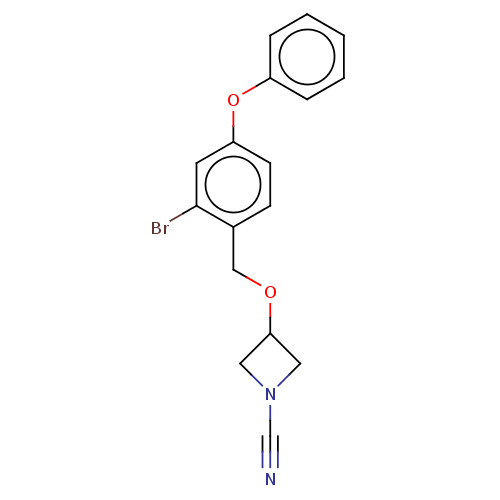
Affinity DataIC50: 0.460nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.470nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.680nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.850nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
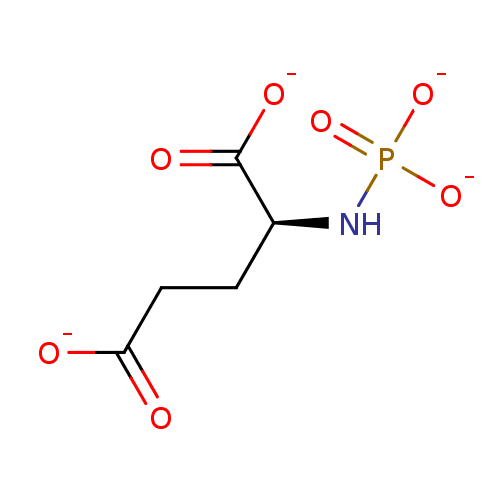
TargetGlutamate carboxypeptidase 2(Homo sapiens (Human))
San Francisco State University
Curated by ChEMBL
San Francisco State University
Curated by ChEMBL
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.880nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.890nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
Affinity DataIC50: 0.900nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 0.940nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as...More data for this Ligand-Target Pair
TargetN-acylethanolamine-hydrolyzing acid amidase(Homo sapiens (Human))
Northeastern University
Curated by ChEMBL
Northeastern University
Curated by ChEMBL
Affinity DataIC50: 1nMAssay Description:Inhibition of activated human NAAA using fluorogenic PAMCA and N-4-methylcoumarin as substrate incubated for 90 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of recombinant human FAAH expressed in Escherichia coli using fluorogenic AAMCA as substrate by fluorimetric assayMore data for this Ligand-Target Pair