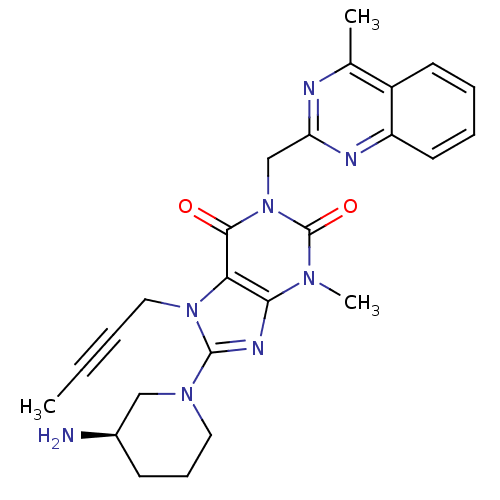
BDBM50228403 (R)-8-(3-aminopiperidin-1-yl)-7-(but-2-ynyl)-3-methyl-1-((4-methylquinazolin-2-yl)methyl)-1H-purine-2,6(3H,7H)-dione::8-[(3R)-3-Aminopiperidin-1-yl]-7-but-2-yn-1-yl-3-methyl-1-[(4-methylquinazolin-2-yl)methyl]-3,7-dihydro-1H-purine-2,6-dione::CHEMBL237500::LINAGLIPTIN::US10202383, Example 2(142)::US10358449, Linagliptin::US20240115577, Compound 5::US9255098, Linagliptin::US9321791, 2(142)::US9556175, 2(142)
SMILES CC#CCn1c(nc2n(C)c(=O)n(Cc3nc(C)c4ccccc4n3)c(=O)c12)N1CCC[C@@H](N)C1
InChI Key InChIKey=LTXREWYXXSTFRX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 59 hits for monomerid = 50228403
Found 59 hits for monomerid = 50228403
Affinity DataKd: 0.00660nMAssay Description:Binding affinity to human recombinant DPP4 (39 to 766 residues) by surface plasmon resonance analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human C-terminal step-tagged DPP4 expressed using baculovirus systemMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human DPP4 preincubated for 30 mins followed by Gly-Pro-AMC addition measured for 50 mins by continuous fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Inhibition of mouse DPP-4 using Gly-Pro-AMC as substrate measured after 5 mins by CF assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.140nMAssay Description:Inhibition of human recombinant DPP-4 (1 to 530 residues) using Gly-Pro-AMC as substrate preincubated for 30 mins followed by substrate addition meas...More data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Human DPP4 activity assay data were obtained using a DPP4 Activity Assay Kit (Sigma-Aldrich, MAK088) according to the manufacturer's instructions...More data for this Ligand-Target Pair
Affinity DataIC50: 0.210nMAssay Description:DPP-IV could hydrolyze Gly-Pro-Aminoluciferin at room temperature to generate Aminoluciferin, which could produce glow type luminescent signals in a ...More data for this Ligand-Target Pair
Affinity DataIC50: 0.320nMAssay Description:The synthetic title compounds 7 (a-j) and 8 (a-j) were screened for in vitro DPP-4 inhibition using DPP-4 activity assay kit (Krishgen BioSystems). D...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:Inhibition of human DPP4 using Gly-Pro-AMC as substrate incubated for 30 mins by continuous fluorescent assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMT: 2°CAssay Description:Gly-Pro-7-amido-4-methylcoumarin can be hydrolyzed by dipeptidyl peptidase IV (DPP-IV) at room temperature, to generate 7-amido-4-methyl coumarin, wh...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:50 μl substrate solution (AFC; AFC is amido-4-trifluoromethylcoumarin), final concentration 100 μM, were placed in black microtitre plates....More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of DPP4 in human Caco-2 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.8Assay Description:50 μl substrate solution (AFC; AFC is amido-4-trifluoromethylcoumarin), final concentration 100 μM, were placed in black microtitre plates....More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human DPP4 in Caco-2 cells by fluorescene assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The DPP-4 Drug Discovery Kit (Enzo Life Sciences International, Inc.) was used for the assay of inhibition of DPP-4 activity. The assay is based on t...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMpH: 7.8Assay Description:50 μl substrate solution (AFC; AFC is amido-4-trifluoromethylcoumarin), final concentration 100 μM, were placed in black microtitre plates. 20 ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of DPP4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of dipeptidyl peptidase 4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of DPP-4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of DPP4 in human plasma using Gly-Pro-AMC as substrate by fluorimetric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human recombinant DPP-4 expressed in human Caco-2 cells using H-Ala-Pro-7-amido-4-trifluoromethylcoumarin as substrate measured after 1...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human recombinant DPP4 (39 to 766 residues) using Ala-Pro-AFC as substrate incubated for 1 hr by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of DPP4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:Inhibition of human DPP4 in pH 7.4 Tris buffer using AP-7-ATFMC as substrate preincubated for 15 mins followed by substrate addition by microplate re...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:Inhibition of human recombinant DPP-4 using (H-Gly-Pro-AMC as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of DPP4 purified from human seminal plasma using Gly-Pro-p-nitroanilide as substrate by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of human DPP4More data for this Ligand-Target Pair
Affinity DataEC50: 3.10nMAssay Description:Inhibition of DPP-4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataKd: 5.30nMAssay Description:Binding affinity to human recombinant DPP4 (39 to 766 residues) at 5 uM by isothermal titration calorimetryMore data for this Ligand-Target Pair
Affinity DataIC50: 71nMAssay Description:Inhibition of FAP (unknown origin) preincubated for 20 mins followed by Nle-Pro-AMC addition measured for 40 mins by continuous fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 89nMAssay Description:Inhibition of FAPalphaMore data for this Ligand-Target Pair
Affinity DataIC50: 295nMAssay Description:Inhibition of M1 receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 295nMAssay Description:Displacement of [N-methyl-3H]scopolamine from human recombinant muscarinic M1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Inhibition of M1 receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:Inhibition of recombinant mouse FAP purified from HEK293 cell supernatant using Ala-Pro-p-nitroanilide as substrate by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:Inhibition of mouse recombinant FAP expressed in HEK293 cells using Ala-Pro-p-nitroanilide as substrate incubated for 15 mins prior to substrate addi...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of DPP9More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of Porphyromonas gingivalis N-terminal His-tagged DPP4 expressed in Escherichia coli using Gly-Pro-p-nitroanilide as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of human ERG by dofetilide binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of DPP8More data for this Ligand-Target Pair
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of human DDP-8 in baculovirus infected Sf9 insect cellsMore data for this Ligand-Target Pair
TargetPOU domain, class 2, transcription factor 1(Human)
Nippon Boehringer Ingelheim
Curated by ChEMBL
Nippon Boehringer Ingelheim
Curated by ChEMBL
Affinity DataIC50: 4.10E+4nMAssay Description:Inhibition of OCT1 (unknown origin) expressed in HEK293 cells assessed as reduction of [14C]metformin substrate uptake at 100 uM by liquid scintillat...More data for this Ligand-Target Pair
TargetPOU domain, class 2, transcription factor 1(Human)
Nippon Boehringer Ingelheim
Curated by ChEMBL
Nippon Boehringer Ingelheim
Curated by ChEMBL
Affinity DataIC50: 4.50E+4nMAssay Description:Inhibition of OCT1 (unknown origin) expressed in HEK293 cells assessed as reduction of [ethyl 1-14C]TEA substrate uptake at 100 uM by liquid scintill...More data for this Ligand-Target Pair
TargetPOU domain, class 2, transcription factor 2(Human)
Nippon Boehringer Ingelheim
Curated by ChEMBL
Nippon Boehringer Ingelheim
Curated by ChEMBL
Affinity DataIC50: 8.00E+4nMAssay Description:Inhibition of OCT2 (unknown origin) expressed in HEK293 cells assessed as reduction of [14C]metformin substrate uptake at 100 uM by liquid scintillat...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of QPP (unknown origin) preincubated for 30 mins followed by Nle-Pro-AMC addition measured for 50 mins by continuous fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human recombinant PREP expressed in Escherichia coli using Z-Gly-Pro-p-nitroanilide as substrate incubated for 15 mins prior to substra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of DPP2 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of DPP9 (unknown origin) preincubated for 20 mins followed by Gly-Pro-AMC addition measured for 50 mins by continuous fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of bovine DPP9More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of DPP2 purified from human seminal plasma using Lys-Ala-p-nitroanilide as substrate by spectrophotometryMore data for this Ligand-Target Pair