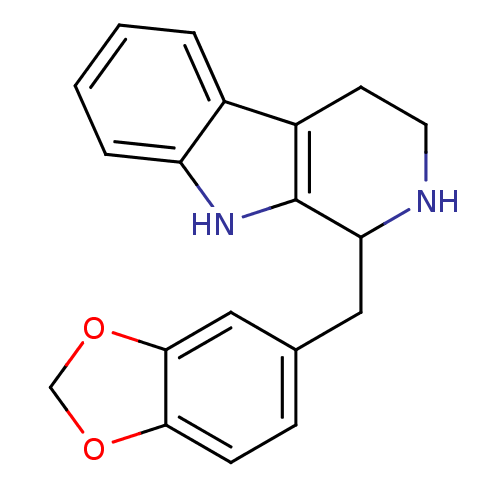
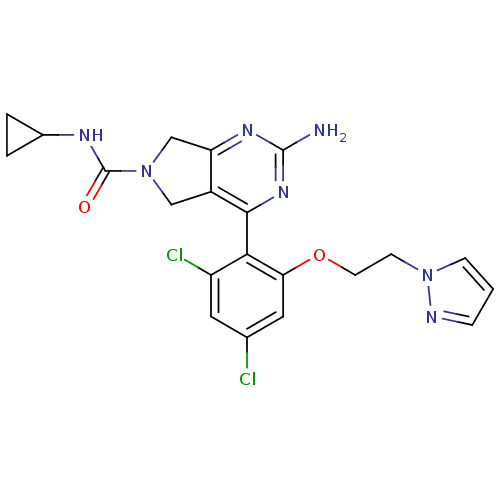
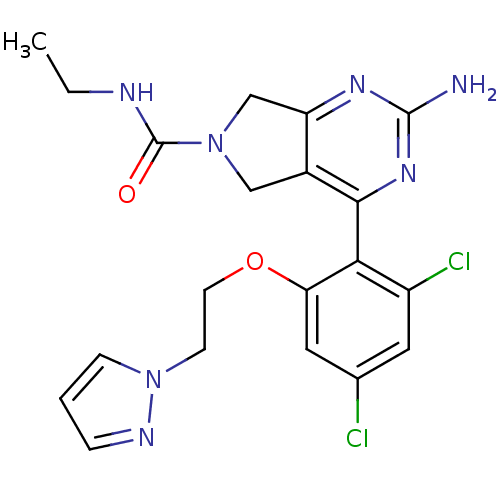
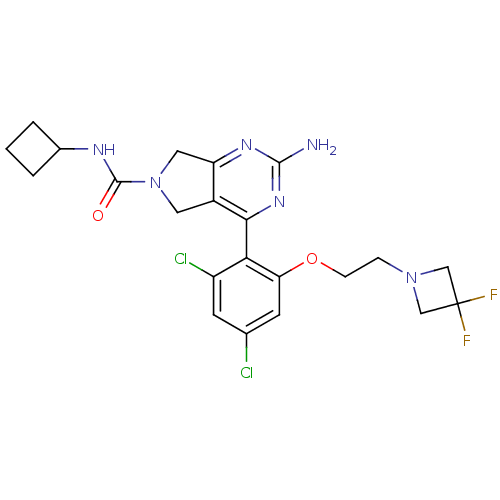
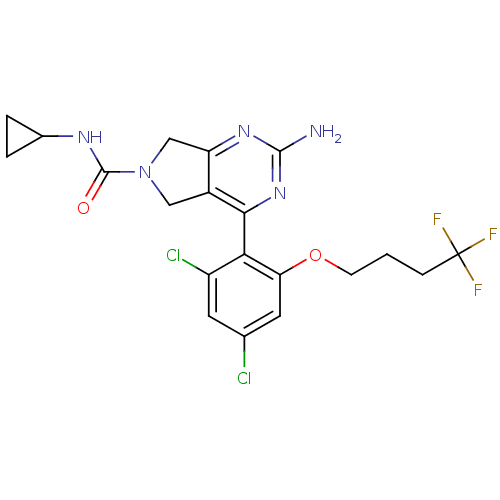
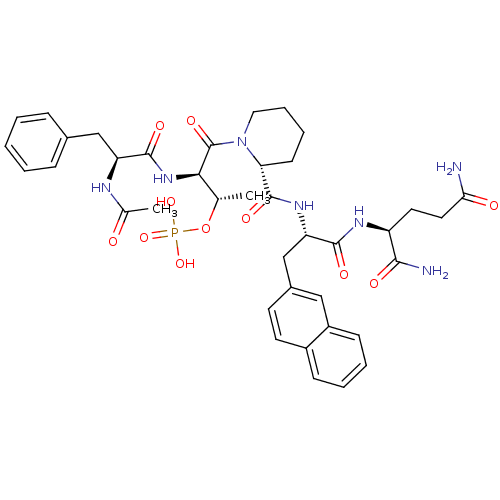
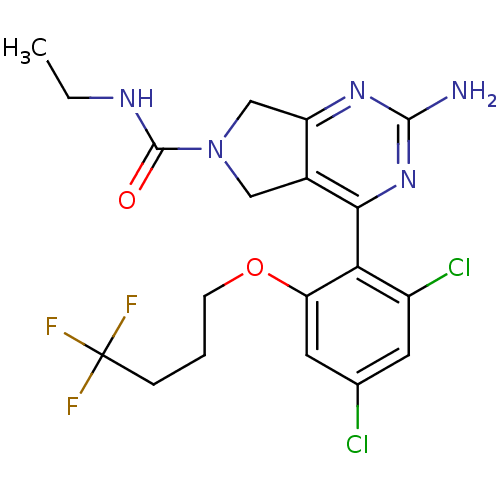
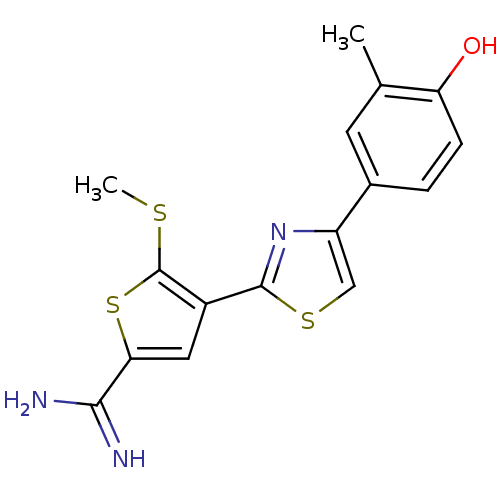
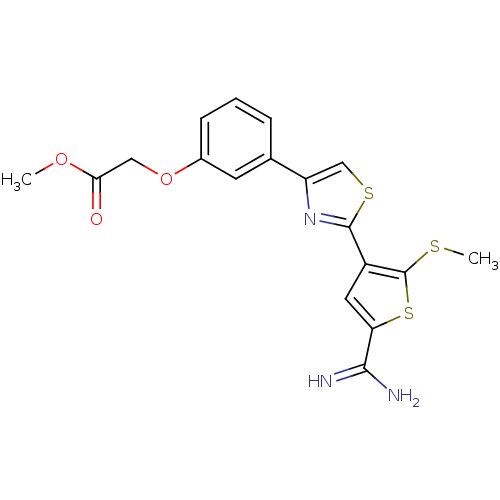
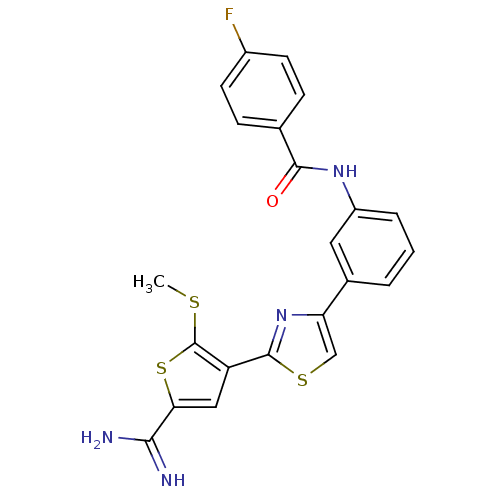
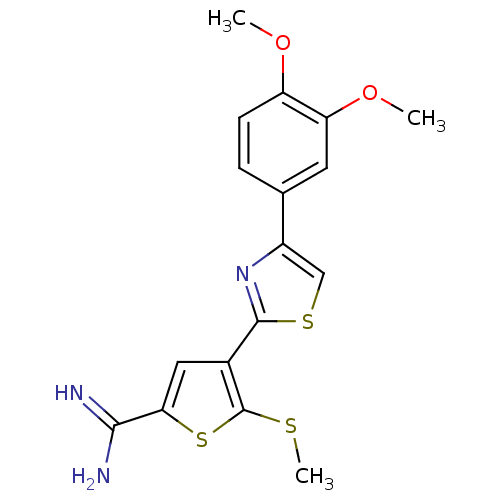
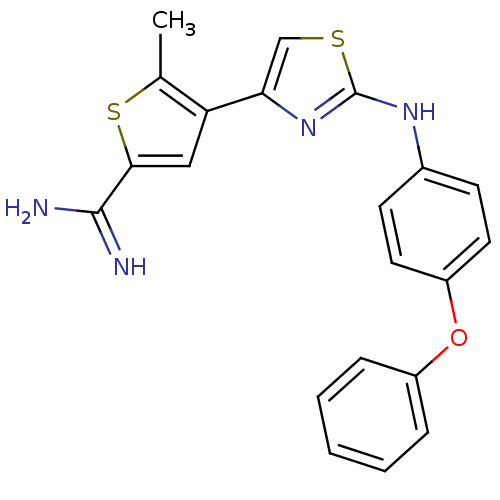
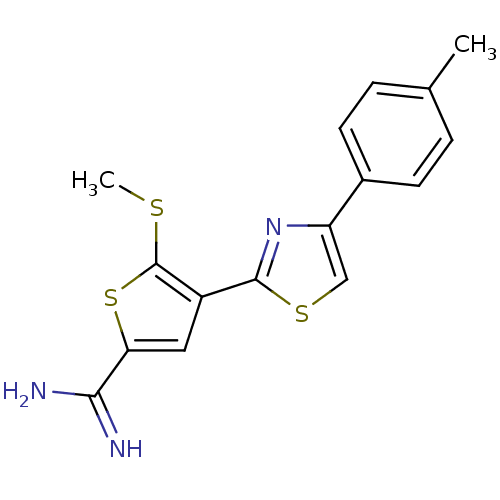
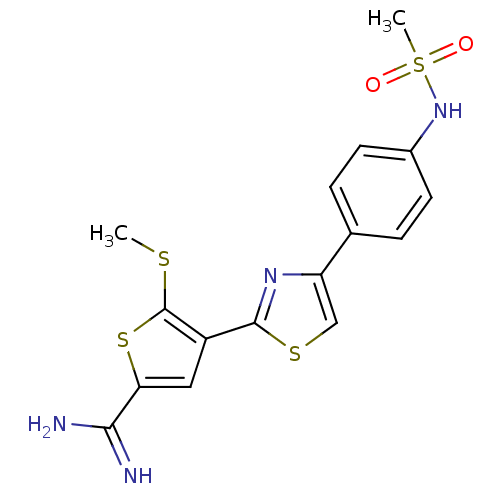
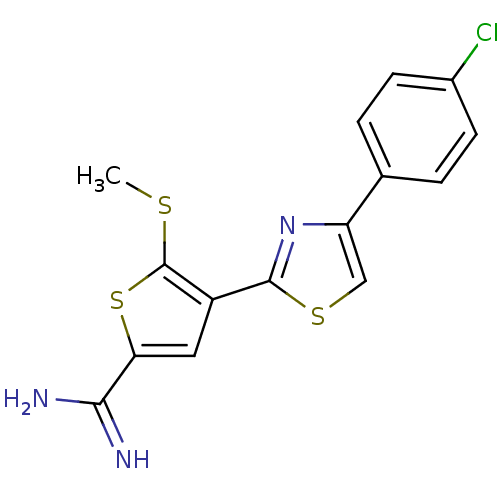
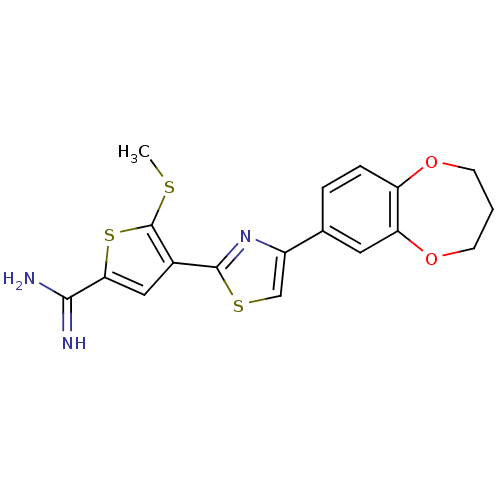
TargetPeptidyl-prolyl cis-trans isomerase NIMA-interacting 1(Homo sapiens (Human))
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Affinity DataKi: 1.20nMAssay Description:Inhibition of human Pin1 PPIase Activity by protease free PPIase assayMore data for this Ligand-Target Pair
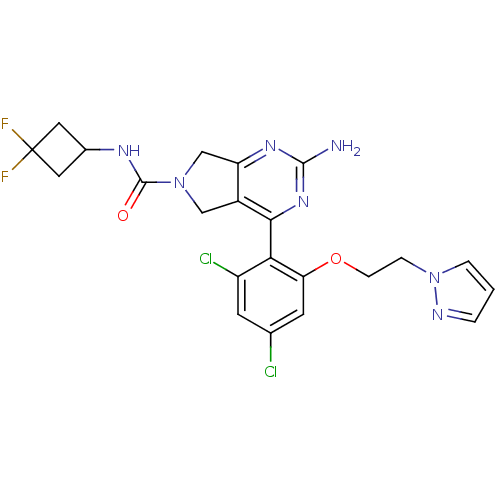
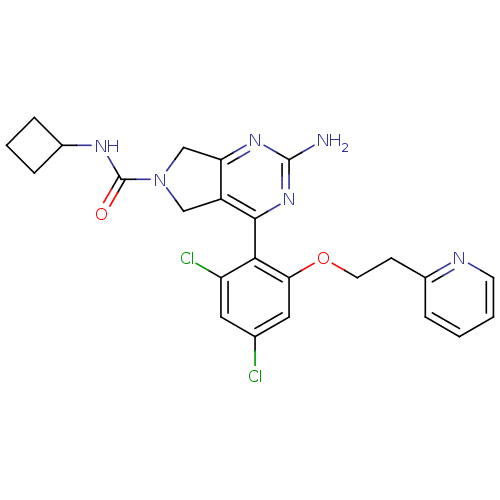
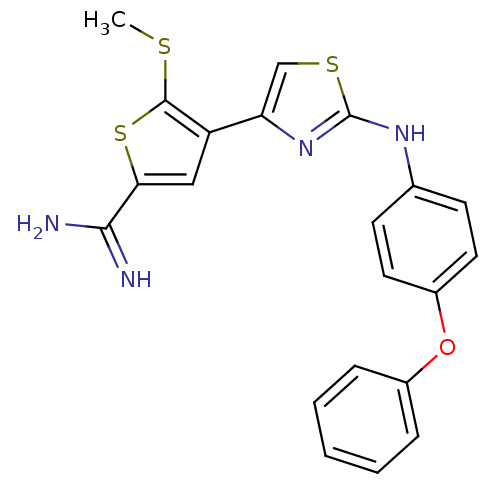
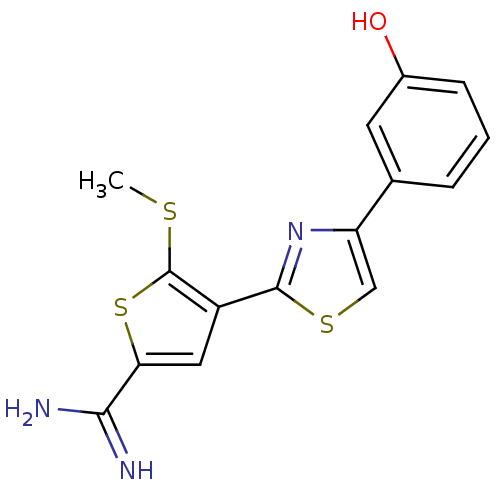
TargetPeptidyl-prolyl cis-trans isomerase NIMA-interacting 1(Homo sapiens (Human))
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Affinity DataKi: 4.80nMAssay Description:Inhibition of human Pin1 PPIase Activity by protease free PPIase assayMore data for this Ligand-Target Pair
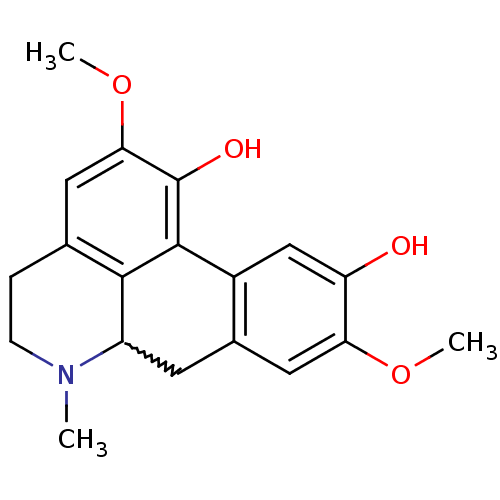
Affinity DataKi: 6nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 7nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
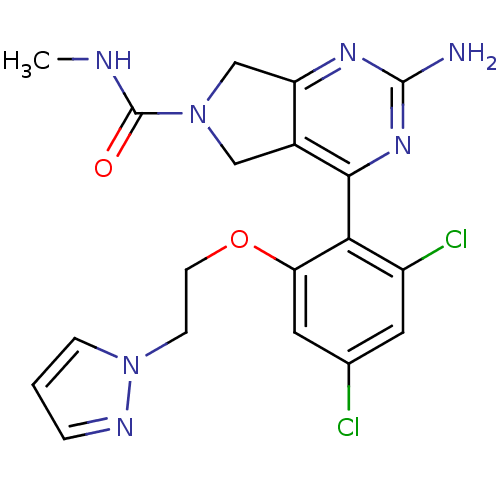
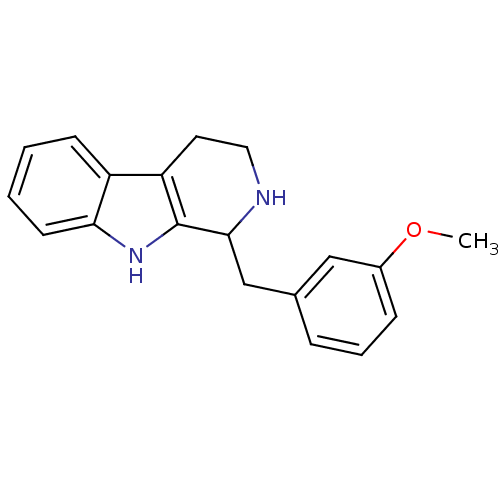
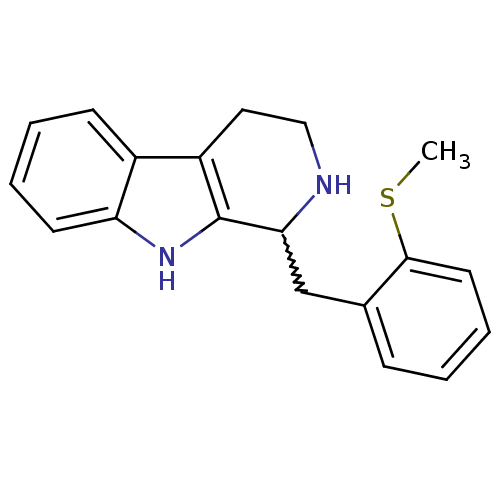
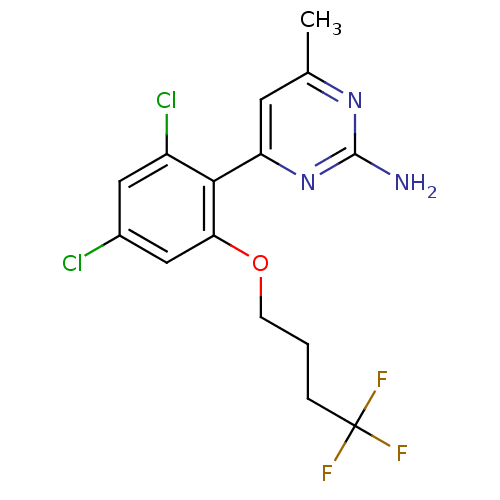
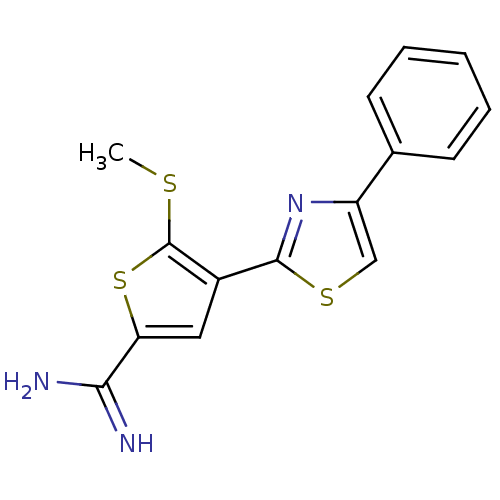
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 9nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 9nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 11nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 12nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
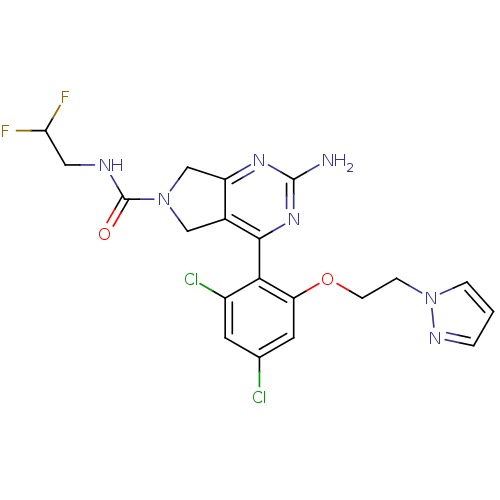
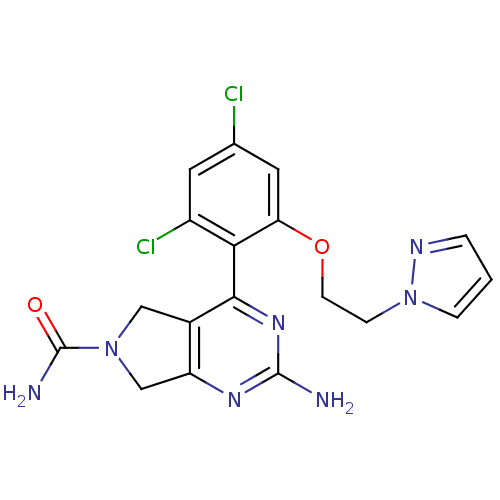
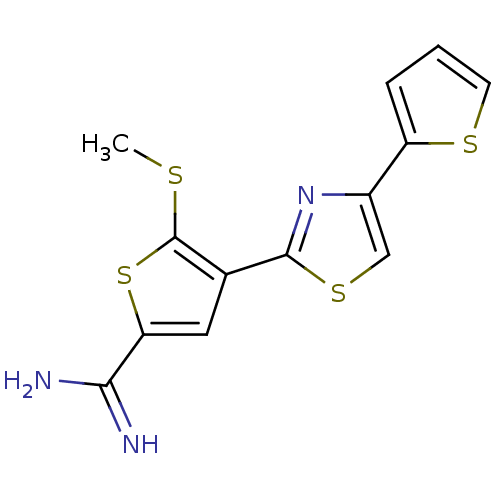
TargetPeptidyl-prolyl cis-trans isomerase NIMA-interacting 1(Homo sapiens (Human))
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Max Planck Research Unit For Enzymology Of Protein Folding
Curated by ChEMBL
Affinity DataKi: 18.3nMAssay Description:Inhibition of human Pin1 PPIase Activity by protease free PPIase assayMore data for this Ligand-Target Pair
Affinity DataKi: 20nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
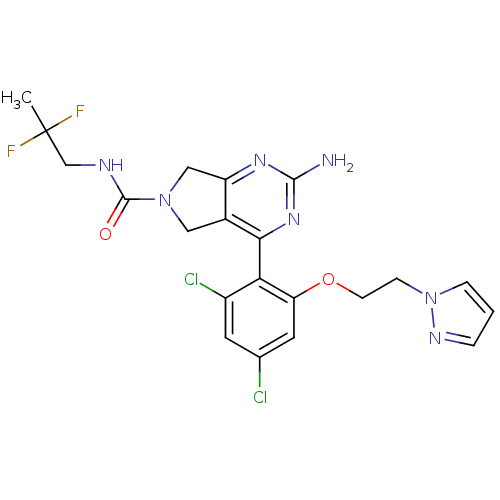
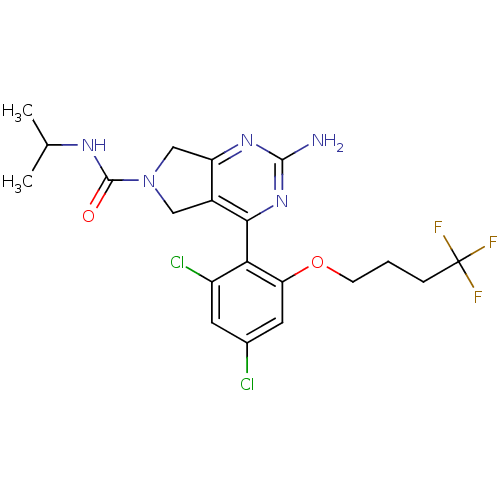
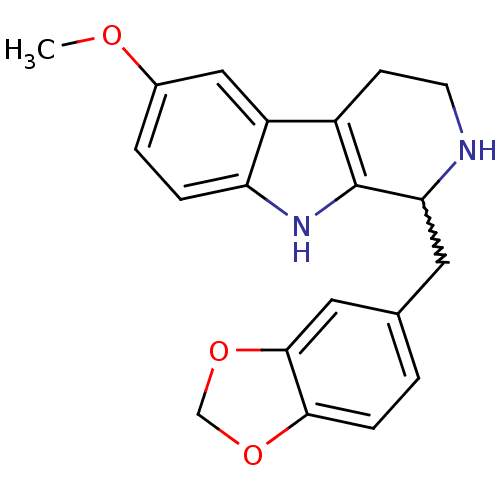
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 20nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 21nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 26nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 26nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 27nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 35nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 35nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 36nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
Affinity DataKi: 40nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
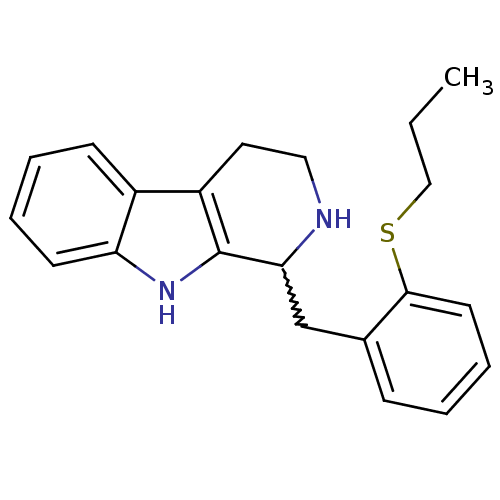
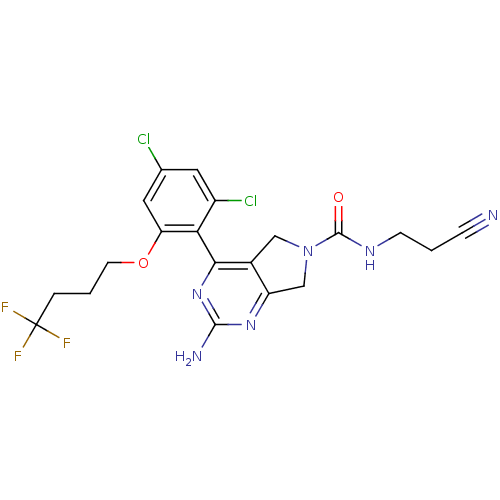
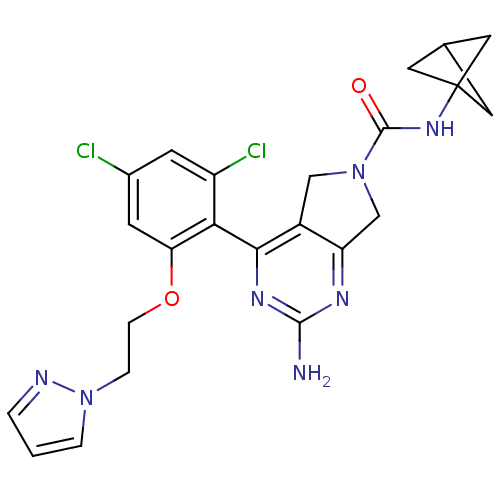
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 44nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
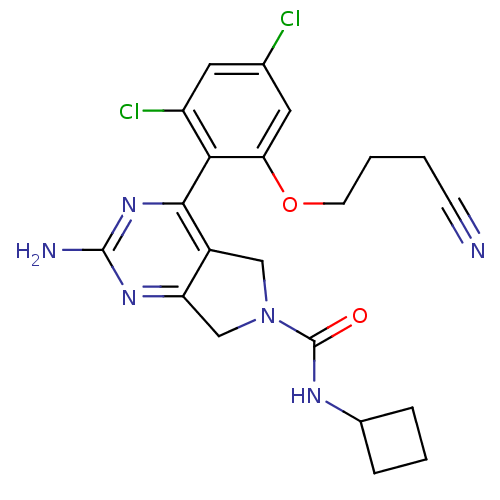
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 44nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 45nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 46nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 47nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Displacement of [3H]-(R)-2-(5-chloro-2,4-dihydroxybenzoyl)-N-ethylisoindoline-1-carboxamide from human his(6)-tagged HSP90alpha after 30 mins by scin...More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 58nMAssay Description:Inhibition of Human kidney cell urokinaseMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 60nMAssay Description:In vitro inhibitory activity against Urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 70nMAssay Description:In vitro inhibitory activity against Urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 80nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 80nMAssay Description:In vitro inhibitory activity against Urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 86nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 89nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 90nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 91nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 94nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 94nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 99nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Homo sapiens (Human))
University Of Strathclyde
Curated by ChEMBL
University Of Strathclyde
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Displacement of [3H]5CT from 5HT7 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 100nMAssay Description:Inhibitory activity against serine protease urokinase-type plasminogen activator (microPa)More data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 101nMAssay Description:Inhibition of Human kidney cell urokinaseMore data for this Ligand-Target Pair
TargetUrokinase-type plasminogen activator(Homo sapiens (Human))
3-Dimensional Pharmaceuticals
Curated by ChEMBL
3-Dimensional Pharmaceuticals
Curated by ChEMBL
Affinity DataKi: 102nMAssay Description:Inhibition of Human kidney cell urokinaseMore data for this Ligand-Target Pair

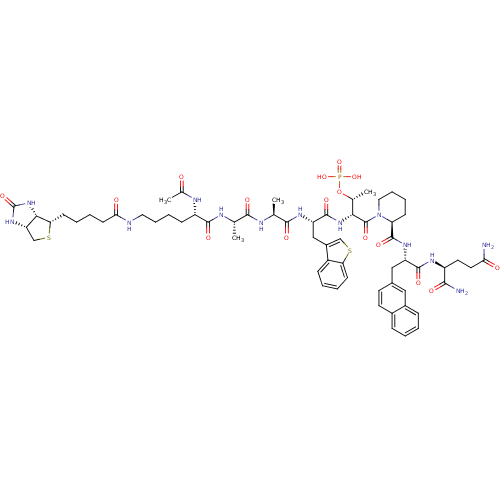
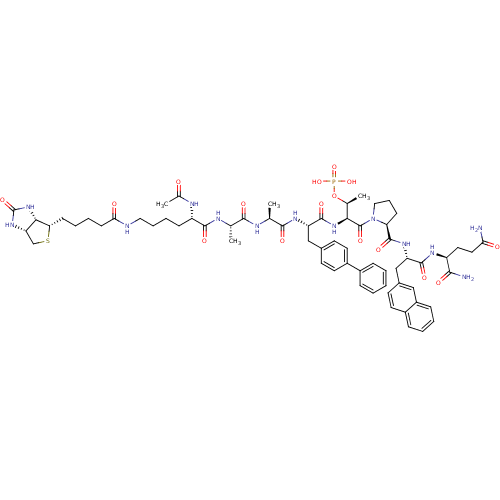
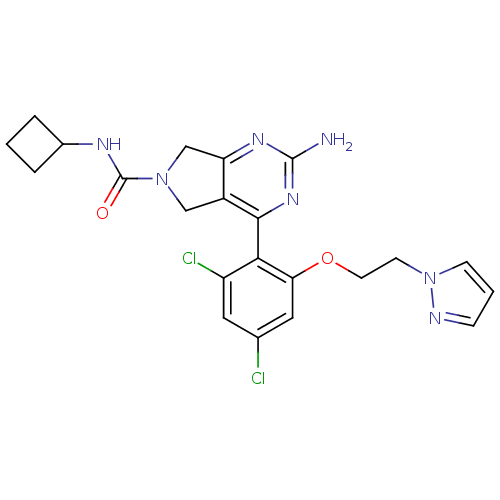
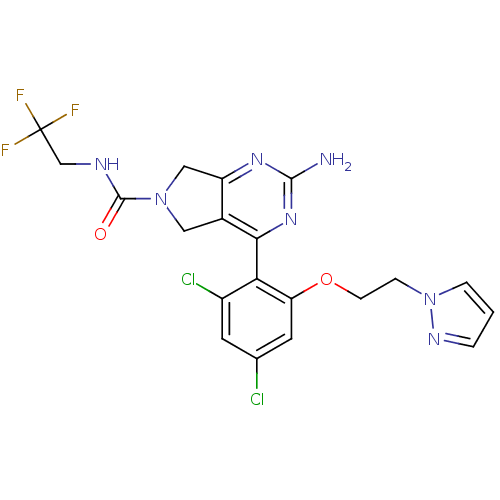
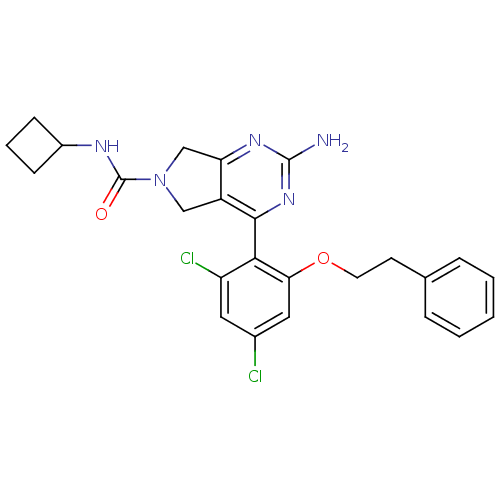
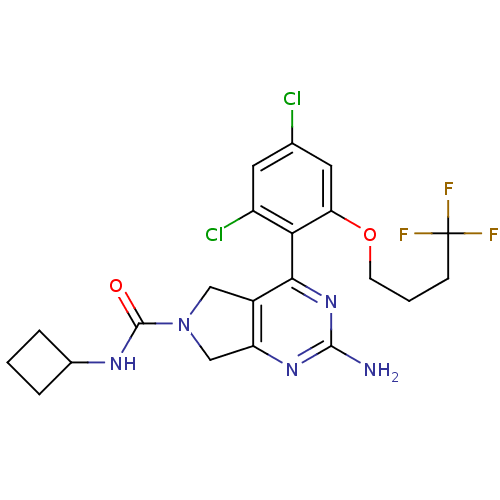
 3D Structure (crystal)
3D Structure (crystal)