Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2C9
Ligand
BDBM50211854
Substrate
n/a
Meas. Tech.
ChEMBL_436453 (CHEMBL904759)
IC50
75000±n/a nM
Citation
 Giblin, GM; O'Shaughnessy, CT; Naylor, A; Mitchell, WL; Eatherton, AJ; Slingsby, BP; Rawlings, DA; Goldsmith, P; Brown, AJ; Haslam, CP; Clayton, NM; Wilson, AW; Chessell, IP; Wittington, AR; Green, R Discovery of 2-[(2,4-dichlorophenyl)amino]-N-[(tetrahydro- 2H-pyran-4-yl)methyl]-4-(trifluoromethyl)- 5-pyrimidinecarboxamide, a selective CB2 receptor agonist for the treatment of inflammatory pain. J Med Chem 50:2597-600 (2007) [PubMed] Article
Giblin, GM; O'Shaughnessy, CT; Naylor, A; Mitchell, WL; Eatherton, AJ; Slingsby, BP; Rawlings, DA; Goldsmith, P; Brown, AJ; Haslam, CP; Clayton, NM; Wilson, AW; Chessell, IP; Wittington, AR; Green, R Discovery of 2-[(2,4-dichlorophenyl)amino]-N-[(tetrahydro- 2H-pyran-4-yl)methyl]-4-(trifluoromethyl)- 5-pyrimidinecarboxamide, a selective CB2 receptor agonist for the treatment of inflammatory pain. J Med Chem 50:2597-600 (2007) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2C9
Synonyms:
(R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase
Type:
Enzyme
Mol. Mass.:
55636.33
Organism:
Homo sapiens (Human)
Description:
P11712
Residue:
490
Sequence:
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
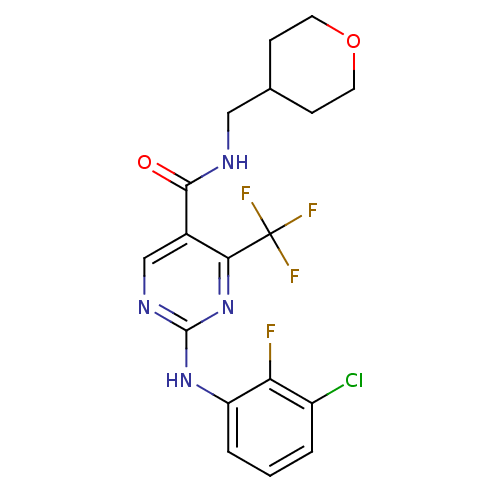
Inhibitor
Name:
BDBM50211854
Synonyms:
2-(2-fluoro-3-chlorophenylamino)-4-trifluoromethylpyrimidine-5-carboxylic acid(tetrahydro-pyran-4-ylmethyl)-amide | CHEMBL225413
Type:
Small organic molecule
Emp. Form.:
C18H17ClF4N4O2
Mol. Mass.:
432.8
SMILES:
Fc1c(Cl)cccc1Nc1ncc(C(=O)NCC2CCOCC2)c(n1)C(F)(F)F