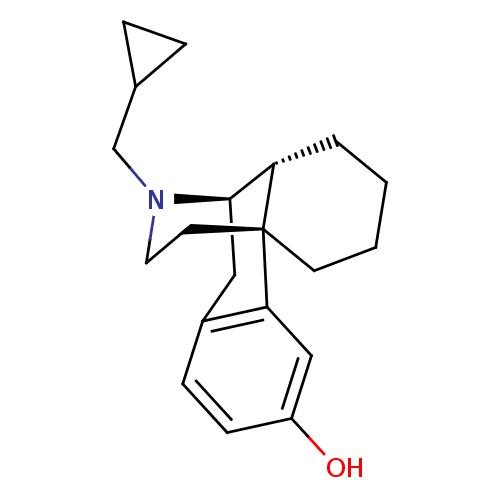
BDBM50135800 (-)-3-Hydroxy-N-cycloproypylmethylmorphinan Mandelate::17-cyclopropylmethyl-(1R,9R,10R)-17-azatetracyclo[7.5.3.01,10.02,7]heptadeca-2(7),3,5-trien-4-ol::3-Hydroxy-N-cyclopropylmethylmorphinan::CHEMBL49269::CYCLORPHAN::US10231963, Table B.6::US10736890, Compound TABLE B.6::US11534436, Compound Table B.6::US9656961, Example 00123
SMILES Oc1ccc2C[C@@H]3[C@@H]4CCCC[C@]4(CCN3CC3CC3)c2c1
InChI Key InChIKey=NLBUEDSBXVNAPB-DFQSSKMNSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50135800
Found 6 hits for monomerid = 50135800
Affinity DataKi: 0.0340nMAssay Description:Displacement of [3H]U-69593 from human kappa opioid receptors expressed in CHO cell membraneMore data for this Ligand-Target Pair
Affinity DataKi: 0.0620nMAssay Description:Displacement of [3H]DAMGO from human mu opioid receptors expressed in CHO cell membraneMore data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Displacement of [3H]naltrindole from human delta opioid receptors expressed in CHO cell membraneMore data for this Ligand-Target Pair
Affinity DataEC50: 0.190nMAssay Description:Agonist activity at huma kappa opioid receptor expressed in CHO cells assessed as U50488-stimulated of [35S]GTP-gamma-S bindingMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:Antagonist activity at human mu opioid receptor expressed in CHO cells assessed as DAMGO-stimulated [35S]GTP-gamma-S bindingMore data for this Ligand-Target Pair
Affinity DataEC50: 0.800nMAssay Description:Agonist activity at human mu opioid receptor expressed in CHO cells assessed as maximal stimulation of [35S]GTP-gamma-S bindingMore data for this Ligand-Target Pair