

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50144945 (2R,3R,4R,5R)-2-Hydroxymethyl-4-methoxy-5-(6-methylamino-purin-9-yl)-tetrahydro-furan-3-ol::(2R,3R,4R,5R)-5-(6-amino-9H-purin-9-yl)-2-(hydroxymethyl)-4-methoxy-tetrahydrofuran-3-ol::2'-O-Methyladenosine::CHEMBL73237
SMILES: CO[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1n1cnc2c(N)ncnc12
InChI Key: InChIKey=FPUGCISOLXNPPC-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine receptor A3 (Human) | BDBM50144945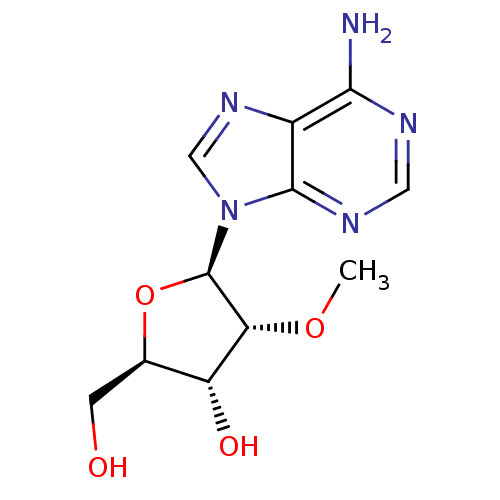 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Human) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Human) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Human) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | n/a | n/a | >3.00E+3 | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| RNA-directed RNA polymerase (Hepatitis C virus genotype 1b (isolate Con1) (HCV)) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | 4.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| RNA-directed RNA polymerase (Hepatitis C virus genotype 1b (isolate Con1) (HCV)) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | n/a | n/a | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rat) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Rat) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transmembrane and immunoglobulin domain-containing 3 (Rat) | BDBM50144945 (CHEMBL73237 | (2R,3R,4R,5R)-5-(6-amino-9H-purin-9-...) | GoogleScholar | UniChem | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||