

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM79212 (2R,3R)-2-(diphenylmethyl)-N-[(2-iodanylphenyl)methyl]-1-azabicyclo[2.2.2]octan-3-amine;ethanedioic acid;hydrate::(2R,3R)-2-(diphenylmethyl)-N-[(2-iodophenyl)methyl]-1-azabicyclo[2.2.2]octan-3-amine;oxalic acid;hydrate::(2R,3R)-2-benzhydryl-N-[(2-iodophenyl)methyl]-1-azabicyclo[2.2.2]octan-3-amine;oxalic acid;hydrate::L-703,606 oxalate salt hydrate::MLS002172464::SMR001254095::[(2R,3R)-2-benzhydrylquinuclidin-3-yl]-(2-iodobenzyl)amine;oxalic acid;hydrate::cid_25102597
SMILES: [H]C12CCN(CC1)[C@H](C(c1ccccc1)c1ccccc1)[C@@H]2NCc1ccccc1I
InChI Key:
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ubiquitin-conjugating enzyme E2 N (Human) | BDBM79212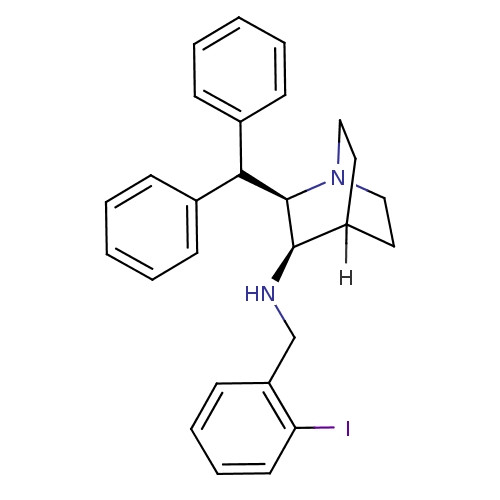 ((2R,3R)-2-(diphenylmethyl)-N-[(2-iodophenyl)methyl...) | GoogleScholar | UniChem | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium channel subfamily K member 9 (Human) | BDBM79212 ((2R,3R)-2-(diphenylmethyl)-N-[(2-iodophenyl)methyl...) | GoogleScholar | UniChem | n/a | n/a | 4.55E+4 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||