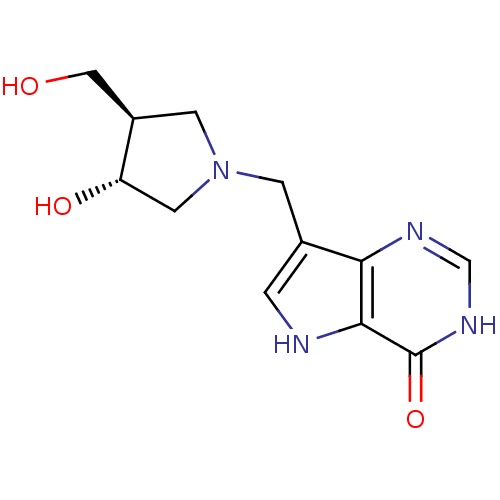
BDBM22109 7-{[(3R,4R)-3-hydroxy-4-(hydroxymethyl)pyrrolidin-1-yl]methyl}-1,5-dihydro-4H-pyrrolo[3,2-d]pyrimidin-4-one::7-{[(3R,4R)-3-hydroxy-4-(hydroxymethyl)pyrrolidin-1-yl]methyl}-1H,4H,5H-pyrrolo[3,2-d]pyrimidin-4-one::CHEMBL1213653::CHEMBL269864::DADMe-ImmH,7
SMILES OC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O
InChI Key InChIKey=AFNHHLILYQEHKK-BDAKNGLRSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 16 hits for monomerid = 22109
Found 16 hits for monomerid = 22109
Affinity DataKi: 0.00900nMAssay Description:Inhibition of human PNP by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0107nMAssay Description:Equilibrium binding affinity to wild type human PNPMore data for this Ligand-Target Pair
Affinity DataKi: 0.0110nMAssay Description:Inhibition of human purine nucleoside phosphorylase by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Inhibition of human His-tagged PNP assessed as equilibrium dissociation constant by measuring reduction in uric acid formation by spectrophotometric ...More data for this Ligand-Target Pair
Affinity DataKi: 0.0160nMAssay Description:Dissociation constant against Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Inhibition of Plasmodium falciparum His-tagged PNP expressed in Escherichia coli assessed as inhibitor constant for enzyme-inhibitor complex formatio...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nM ΔG°: -12.6kcal/molepH: 7.7 T: 2°CAssay Description:PNP activity was monitored by absorbance change in a coupled assay. In the assay, inosine was converted to hypoxanthine, and then hypoxanthine was co...More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Inhibition of Plasmodium falciparum purine nucleoside phosphorylase assessed as slow onset inhibition constant by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 1nM ΔG°: -12.1kcal/molepH: 7.7 T: 2°CAssay Description:PNP activity was monitored by absorbance change in a coupled assay. In the assay, inosine was converted to hypoxanthine, and then hypoxanthine was co...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Binding affinity towards Human Purine Nucleoside Phosphorylase was reportedMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Inhibition of human purine nucleoside phosphorylase assessed as slow onset inhibition constant by xanthine-oxidase coupled assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Inhibition of human PNPMore data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Inhibition of human His-tagged PNP assessed as inhibitor constant for enzyme-inhibitor complex formation by measuring reduction in uric acid formatio...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nM ΔG°: -12.1kcal/molepH: 7.7 T: 2°CAssay Description:PNP activity was monitored by absorbance change in a coupled assay. In the assay, inosine was converted to hypoxanthine, and then hypoxanthine was co...More data for this Ligand-Target Pair
Affinity DataKi: 1.10nMAssay Description:Initial binding affinity to wild type human PNPMore data for this Ligand-Target Pair
Affinity DataKd: 0.00850nMAssay Description:Binding affinity to human PNPMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)