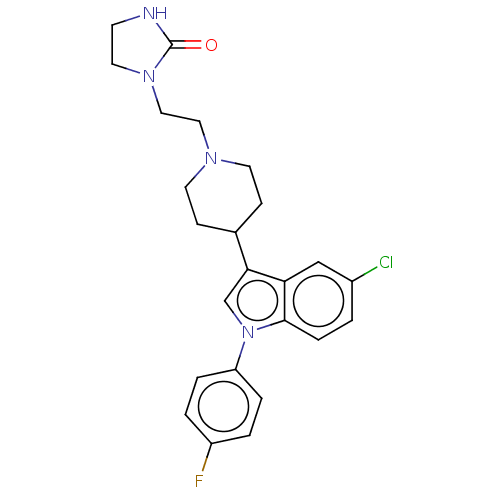
BDBM50001786 1-(2-{4-[5-chloro-1-(4-fluorophenyl)-1H-indol-3-yl]piperidin-1-yl}ethyl)imidazolidin-2-one::1-[2-[4-[5-chloro-1-(4-fluorophenyl)-indol-3-yl]-1-piperidyl]ethyl]imidazolidin-2-one::CHEMBL12713::Sertindole
SMILES Fc1ccc(cc1)-n1cc(C2CCN(CCN3CCNC3=O)CC2)c2cc(Cl)ccc12
InChI Key InChIKey=GZKLJWGUPQBVJQ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 174 hits for monomerid = 50001786
Found 174 hits for monomerid = 50001786
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]ketanserin (0.5 nM) from rat cerebral cortex 5-hydroxytryptamine 2A receptorsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Affinity DataKi: 0.200nMAssay Description:Displacement of [3H]prozosin from human cloned 5HT2C receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Affinity DataKi: 0.330nMAssay Description:Displacement of [3H]prazosin (0.5 nM) from hamster Alpha-1B adrenergic receptor expressed in rat-1 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.330nMAssay Description:Displacement of [3H]prozosin from hamster cloned alpha1b receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.330nMAssay Description:Displacement of [3H]prazosine from hamster alpha1B adrenoceptor expressed in Rat1 cells after 20 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.370nMAssay Description:Displacement of [3H]prazosine from bovine alpha1A adrenoceptor expressed in BHK cells after 20 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.370nMAssay Description:Displacement of [3H]prazosin (0.3 nM) from bovine Alpha-1A adrenergic receptor expressed in BHK cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.370nMAssay Description:Displacement of [3H]prozosin from bovine cloned alpha1a receptor expressed in BHK cellsMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Rattus norvegicus (rat))
University of Reading
Curated by PDSP Ki Database
University of Reading
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
TargetD(2) dopamine receptor(Rattus norvegicus (rat))
University of Reading
Curated by PDSP Ki Database
University of Reading
Curated by PDSP Ki Database
TargetD(2) dopamine receptor(Rattus norvegicus (rat))
University of Reading
Curated by PDSP Ki Database
University of Reading
Curated by PDSP Ki Database
Affinity DataKi: 0.450nMAssay Description:Displacement of [3H]spiperone (0.5 nM) from rat corpus striatum dopamine D2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 0.450nMAssay Description:Displacement of [3H]spiperone from human dopamine D2 receptor expressed in CHO cells after 30 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.450nMAssay Description:Displacement of [3H]prozosin from human cloned dopamine D2 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Rattus norvegicus (rat))
University of Reading
Curated by PDSP Ki Database
University of Reading
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2C(Rattus norvegicus (Rat))
H. Lundbeck
Curated by PDSP Ki Database
H. Lundbeck
Curated by PDSP Ki Database
Affinity DataKi: 0.510nMAssay Description:Displacement of [3H]prozosin from human cloned histamine H1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Rattus norvegicus (Rat))
H. Lundbeck
Curated by PDSP Ki Database
H. Lundbeck
Curated by PDSP Ki Database
Affinity DataKi: 0.510nMAssay Description:Displacement of [3H]mesulergine (0.5 nM) from rat 5-hydroxytryptamine 2C receptor expressed in SR-3T3 cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Affinity DataKi: 0.600nMAssay Description:Binding affinity towards Serotonin 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Affinity DataKi: 0.660nMAssay Description:Displacement of [3H]prazosine from rat alpha1D adrenoceptor expressed in CHO cells after 20 mins by scintillation counting analysisMore data for this Ligand-Target Pair
Affinity DataKi: 0.660nMAssay Description:Displacement of [3H]prazosin (0.3 nM) from rat Alpha-1D adrenergic receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataKi: 0.660nMAssay Description:Displacement of [3H]prozosin from rat cloned alpha1d receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Target5-hydroxytryptamine receptor 2A(Homo sapiens (Human))
Mayo Foundation
Curated by PDSP Ki Database
Mayo Foundation
Curated by PDSP Ki Database
Affinity DataKi: 0.850nMAssay Description:Binding affinity towards human serotonin 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Affinity DataKi: 1nMAssay Description:Displacement of [3H]mesulergine from human 5HT2C receptor in human tsA201 cellsMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2C(Homo sapiens (Human))
University Of Copenhagen
Curated by ChEMBL
University Of Copenhagen
Curated by ChEMBL
Affinity DataKi: 1.30nMAssay Description:Binding affinity towards human serotonin 5-hydroxytryptamine 2C receptorMore data for this Ligand-Target Pair
TargetD(3) dopamine receptor(Rattus norvegicus (Rat))
University of Reading
Curated by PDSP Ki Database
University of Reading
Curated by PDSP Ki Database
TargetAlpha-1A/Alpha-1B/Alpha-1D adrenergic receptor(Homo sapiens (Human))
Prestwick Chemical
Curated by ChEMBL
Prestwick Chemical
Curated by ChEMBL
Affinity DataKi: 1.80nMAssay Description:Inhibition of currents elicited by GABA at EC5 (6-12 uM)More data for this Ligand-Target Pair
TargetAlpha-1A adrenergic receptor(Homo sapiens (Human))
Case Western Reserve University
Curated by PDSP Ki Database
Case Western Reserve University
Curated by PDSP Ki Database