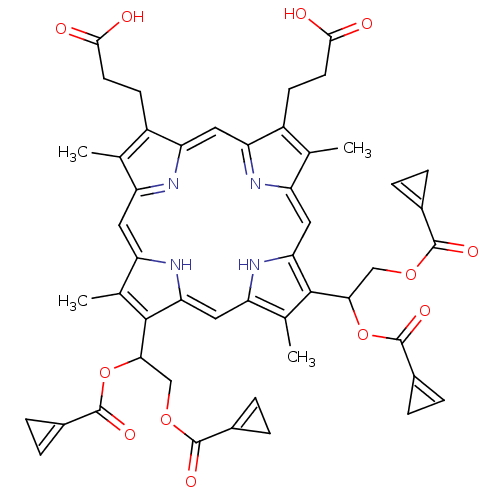
BDBM50004778 3-[18-(2-Carboxy-ethyl)-8,13-bis-(1,2-diacetoxy-ethyl)-3,7,12,17-tetramethyl-22,24-dihydro-porphin-2-yl]-propionic acid::CHEMBL326831
SMILES Cc1c(CCC(O)=O)c2cc3nc(cc4[nH]c(cc5[nH]c(cc1n2)c(C)c5C(COC(=O)C1=CC1)OC(=O)C1=CC1)c(C)c4C(COC(=O)C1=CC1)OC(=O)C1=CC1)c(C)c3CCC(O)=O
InChI Key InChIKey=PFJCIZMGZQBSQR-QGQUFCMSSA-N
Data 12 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 12 hits for monomerid = 50004778
Found 12 hits for monomerid = 50004778
Affinity DataIC50: 4.00E+3nMAssay Description:Inhibitory concentration against pepsinMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Inhibition of HIV-2 protease in 5%DMSOMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibitory concentration against cathepsin DMore data for this Ligand-Target Pair
Affinity DataIC50: 230nMAssay Description:Inhibitory concentration against HIV-2 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibitory concentration against reninMore data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Inhibitory concentration against HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University Of California
Curated by ChEMBL
University Of California
Curated by ChEMBL
Affinity DataIC50: 185nMAssay Description:Inhibition of HIV-1 protease in 5%DMSOMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University Of California
Curated by ChEMBL
University Of California
Curated by ChEMBL
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibitory concentration against HIV-1 protease in the absence of DMSOMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University Of California
Curated by ChEMBL
University Of California
Curated by ChEMBL
Affinity DataIC50: >1.50E+5nMAssay Description:Binding affinity against HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University Of California
Curated by ChEMBL
University Of California
Curated by ChEMBL
Affinity DataIC50: 50nMAssay Description:Inhibitory concentration against HIV-1 protease in the absence of DMSOMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Inhibition of HIV-2 protease in 5%DMSOMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
University Of California
Curated by ChEMBL
University Of California
Curated by ChEMBL
Affinity DataIC50: 185nMAssay Description:Inhibitory concentration against HIV-1 protease in the absence of DMSOMore data for this Ligand-Target Pair