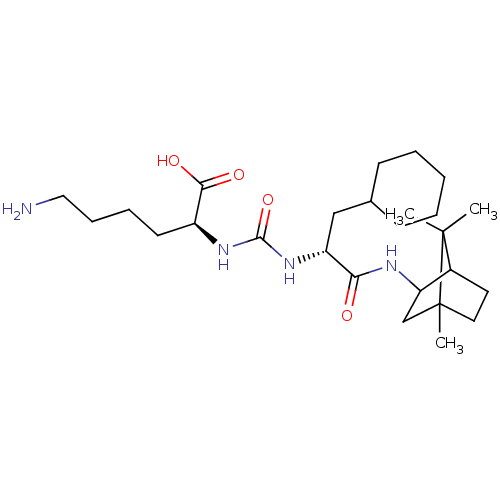
BDBM50089684 CHEMBL3577447
SMILES Cl.CC1(C)C2CCC1(C)CC2NC(=O)[C@@H](CC1CCCCC1)NC(=O)N[C@@H](CCCCN)C(O)=O
InChI Key InChIKey=ZZFWSQSQNZHVSP-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50089684
Found 3 hits for monomerid = 50089684
Affinity DataIC50: 6.50E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using midazolam substrate incubated for 10 mins by LC-MS/MS methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+4nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using testosterone substrate incubated for 30 mins by LC-MS/MS methodMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Institute For Infection Research
Curated by ChEMBL
Institute For Infection Research
Curated by ChEMBL
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of human ERG expressed in CHO cells by whole cell patch clamp assayMore data for this Ligand-Target Pair