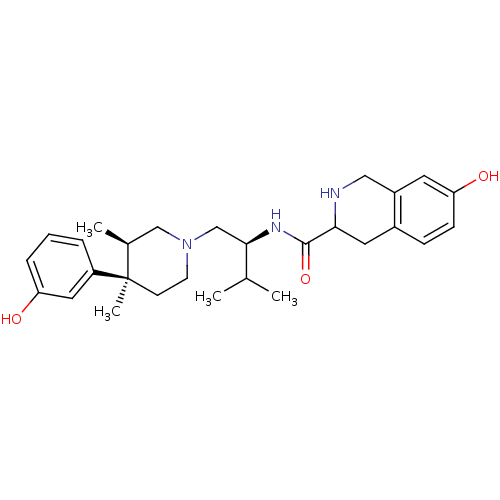
BDBM50102711 7-Hydroxy-1,2,3,4-tetrahydro-isoquinoline-3-carboxylic acid {1-[4-(3-hydroxy-phenyl)-3,4-dimethyl-piperidin-1-ylmethyl]-2-methyl-propyl}-amide::CHEMBL51204
SMILES CC(C)[C@@H](CN1CC[C@](C)([C@@H](C)C1)c1cccc(O)c1)NC(=O)C1Cc2ccc(O)cc2CN1
InChI Key InChIKey=ZLVXBBHTMQJRSX-HUUIFEGFSA-N
Data 9 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 50102711
Found 9 hits for monomerid = 50102711
Affinity DataKi: 0.00600nMAssay Description:Antagonist activity on agonist (U50,488) stimulated [35S]GTP-gamma-S, binding in cloned opioid receptor kappa 1More data for this Ligand-Target Pair
TargetKappa-type opioid receptor(Cavia porcellus (domestic guinea pig))
Research Triangle Institute
Curated by ChEMBL
Research Triangle Institute
Curated by ChEMBL
Affinity DataKi: 0.0200nMAssay Description:Inhibition of stimulation of [35S]GTP-gamma-S, binding produced by the selective agonist (U69593, kappa-receptor), in guinea pig caudate membranes.More data for this Ligand-Target Pair
TargetKappa-type opioid receptor(Cavia porcellus (domestic guinea pig))
Research Triangle Institute
Curated by ChEMBL
Research Triangle Institute
Curated by ChEMBL
Affinity DataKi: 0.320nMAssay Description:Binding affinity to opioid receptor kappa 1 of guinea pig brain, using [3H]U-69593 as radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 2.20nMAssay Description:Inhibition of stimulation of [35S]GTP-gamma-S, binding produced by the selective agonist (DAMGO, mu-receptor), in guinea pig caudate membranes.More data for this Ligand-Target Pair
Affinity DataKi: 3.40nMAssay Description:Antagonist activity on agonist (DAMGO) stimulated [35S]GTP-gamma-S, binding in cloned mu opioid receptors.More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Binding affinity to mu-opioid receptor of rat brain using [3H]DAMGO as radioligand.More data for this Ligand-Target Pair
Affinity DataKi: >100nMAssay Description:Antagonist activity on agonist (SNC-80) stimulated [35S]GTP-gamma-S, binding in cloned opioid receptor mu1More data for this Ligand-Target Pair
Affinity DataKi: >300nMAssay Description:Inhibition of stimulation of [35S]GTP-gamma-S, binding produced by the selective agonist (SNC-80, delta-receptor), in guinea pig caudate membranesMore data for this Ligand-Target Pair
TargetDelta-type opioid receptor(Rattus norvegicus (rat))
Research Triangle Institute
Curated by ChEMBL
Research Triangle Institute
Curated by ChEMBL
Affinity DataKi: 301nMAssay Description:Binding affinity to delta-opioid receptor of rat brain using [3H]DADLE as radioligandMore data for this Ligand-Target Pair